ABILHAND Assessment
Original Editors - Alex Benham
Top Contributors - Alex Benham, Rucha Gadgil and Ashmita Patrao
Objective[edit | edit source]
The ABILHAND is an interview-based assessment of a patient-reported measure of the perceived difficulty in using their hand to perform manual activities in daily activities. The assessment considers the active function of the upper limbs and measures the ability to perform bimanual tasks, regardless of the way in which these tasks are carried out.[1] All of the versions of the assessment are available for from the following website[2] and contain all of the assessment information, downloads of the pack and also the link to the Rasch analysis.
There are currently versions specifically validated for
Chronic stroke, Rheumatoid Arthritis, Systemic sclerosis, Hand Surgery
These test packs are also available in many languages such as English, French, Dutch, Italian, Swedish, Brazilian-Portuguese, Chinese, Canadian English, Canadian French, Czech, Hungarian, Polish, Russian, Serbian, Spanish, UK English, Spanish USA, Bulgarian, Austrian German, Swiss German, Swiss-French, Norwegian, Slovak, Argentinian Spanish, Mexican Spanish.
Instructions[edit | edit source]
This is an interview-based assessment where the patient is asked to estimate the ease or difficulty of performing a list of activities when carried out without assistance. This can involve any strategy used to carry out the activity, and is self-report and not a physical demonstration of the activity.
The questionnaire is downloaded from the website and one of the 10 random orders of questions. These are read to the patient and scored as either "impossible", "difficult" or "easy". If a task has not been attempted in the last 3 months then it is marked as N/A.
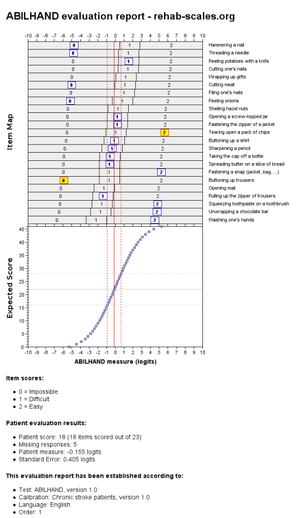
Scoring & Interpretation[edit | edit source]
Analysis of the answers is via a Rasch model of online analysis, which converts the raw scores into a linear measure.
The output of the online analysis is presented as the patient's manual ability (indicated by a red line) alongside the 95% confidence interval. The better the patient's ability, the more the line will be located on the right.
From the output scoresheet, items are ordered from the easiest task at the bottom, to the hardest task at the top. The most likely score according to the difficulty is given and changes according to the patient's manual ability. This allows the score to be used for treatment planning as easier tasks should have recovered first. Outliers or unexpected findings are also noted. See Fig 1.
Summary of score versions[edit | edit source]
ABILHAND in chronic stroke patients[edit | edit source]
- 23 bimanual activities.
- Each item is answered on a 3-level scale (impossible, difficult, easy).
- The item difficulty increases with bimanual involvement.
- Measurement range: approx. 7 logits.
- Measurement error: 0.36 logits in the centre of the scale.
- Least measurable difference: 0.13 logit in the centre of the scale.
- Separation reliability: 0.90 in our sample of 103 chronic stroke patients.
- Invariant item hierarchy across gender, age, the side affected, delay since CVA, level of depression, overall ability in our sample of 103 chronic stroke patients.
ABILHAND in rheumatoid arthritis patients[edit | edit source]
- 27 bimanual activities.
- Each item is answered on a 3-level scale (impossible, difficult, easy).
- The item difficulty increases with constraints in the upper limb joints.
- Measurement range: approx. 8 logits.
- Measurement error: 0.42 logits in the centre of the scale.
- Least measurable difference: 0.15 logit in the centre of the scale.
- Separation reliability: 0.95 in our sample of 112 patients with rheumatoid arthritis.
- Invariant item hierarchy across: gender, age, disease duration, number of DMARDs, tender and swollen joint counts, number of hands affected, DAS score, HAQ score, overall ability and time in our sample of 112 patients with rheumatoid arthritis.
ABILHAND in systemic sclerosis patients[edit | edit source]
- 26 uni- and bimanual activities.
- Each item is answered on a 3-level scale (impossible, difficult, easy).
- Measurement range: approx. 9 logits.
- Measurement error: 0.41 logits in the centre of the scale.
- Least measurable difference: 0.18 logit in the centre of the scale.
- Separation reliability: 0.96 in our sample of 156 patients with systemic sclerosis.
- Invariant item hierarchy across: gender, age, language community, systemic sclerosis subsets, disease duration from the onset of Raynaud phenomenon, Finger-to-palm distance, Total Skin Score, presence of digital pit scars and digital ulcers, score of Health assessment Questionnaire (HAQ), disease activity score and total disease severity score (Total DSS)
- Test-retest reliability: ICC = 0.96 after a delay of 1 month
- Construct validity: ABILHAND vs HAQ (ρ = -0.73), and vs Total DSS(ρ = -0.53).
ABILHAND in hand surgery[edit | edit source]
- 22 bimanual activities, one uni-manual activity
- Each item is answered on a 3-level scale (impossible, difficult, easy).
- The item difficulty increases with bimanual involvement.
- Measurement range: approx. 9 logits.
- Measurement error: 0.38 logits in the centre of the scale.
- Least measurable difference: 0.15 logit in the centre of the scale.
- Separation reliability: 0.90 in our sample of 305 hand surgery patients.
- Invariant item hierarchy across: gender, age, dominant hand involvement, level of education, diagnosis, follow-up.
- Construct validity: ABILHAND vs QuickDASH (ρ = -0.77), vs Numerical pain scale (ρ = -0.49), vs SF-12 PCS (ρ = 0.56) and vs SF-12 MCS (ρ = 0.31).
Evidence
Evaluation of functional outcome measures for the hemiparetic upper limb: a systematic review[4]
Reliability and Validity of the Turkish Version of the ABILHAND Questionnaire in Rheumatoid Arthritis Individuals, Based on Rasch Analysis[5]
Addressing the targeting range of the ABILHAND-56 in relapsing-remitting multiple sclerosis: A mixed methods psychometric study[6]
The ABILHAND questionnaire as a measure of manual ability in chronic stroke patients: Rasch-based validation and relationship to upper limb impairment[7]
References[edit | edit source]
- ↑ Ashford, Stephen, Mike Slade, Fabienne Malaprade, and Lynne Turner-Stokes. "Evaluation of functional outcome measures for the hemiparetic upper limb: a systematic review." Journal of rehabilitation medicine 40, no. 10 (2008): 787-795.
- ↑ http://rssandbox.iescagilly.be/abilhand.html
- ↑ http://rssandbox.iescagilly.be/abilhand-instructions.html
- ↑ Ashford S, Slade M, Malaprade F, Turner-Stokes L. Evaluation of functional outcome measures for the hemiparetic upper limb: a systematic review. Journal of rehabilitation medicine. 2008 Nov 5;40(10):787-95.
- ↑ Basakci Calik B, Gur Kabul E, Tasci M, Erel S, Simsek E, Demir P, Cobankara V. Reliability and Validity of the Turkish Version of the ABILHAND. 2019 Arch Rheumatol 26;34(4)395-405
- ↑ Cleanthous S, Strzok S, Pompilus F, Cano S, Marquis P, Cohan S, Goldman MD, Kresa-Reahl K, Petrillo J, Castrillo-Viguera C, Cadavid D. Addressing the targeting range of the ABILHAND-56 in relapsing–remitting multiple sclerosis: A mixed methods psychometric study. Multiple Sclerosis Journal–Experimental, Translational and Clinical. 2018 May;4(2):2055217318776990.
- ↑ Penta M, Tesio L, Arnould C, Zancan A, Thonnard JL. The ABILHAND questionnaire as a measure of manual ability in chronic stroke patients: Rasch-based validation and relationship to upper limb impairment. Stroke. 2001 Jul;32(7):1627-34.






