Constraint Induced Movement Therapy
This article is currently under review and may not be up to date. Please come back soon to see the finished work! (7/12/2023)
What is Constraint-Induced Movement Therapy or CIMT?[edit | edit source]
Constraint-Induced Movement Therapy (CIMT), also known as CI, is a "rehabilitative strategy". It is aimed at improving the functional use of an affected extremity for those who are impacted by stroke or other neurological conditions. It uses principles of mass practice while restraining the neurologically stronger limb.[1]It has also been defined as a behavioural approach to neurorehabilitation, making use of simple behavioral techniques - shaping being a predominant theme.[2]
CIMT has been described as including the essential components of rehabilitation following neurological injury, which comprise of:[2]
- Repetition - task orientated in manner.
- Constraining of patients, so as to induce use of the impaired limb or function.
- Application of a "package of behavioural methods" (pg 1, para 1) which allow the transference of skills learned in the clinical settings to that of the real-world environment.
While initially developed for the stroke population, it is also being used in populations affected by cerebral palsy, traumatic brain injury, phantom limb pain, focal hand dystonia in musicians and multiple sclerosis.[3]
History of CIMT[edit | edit source]
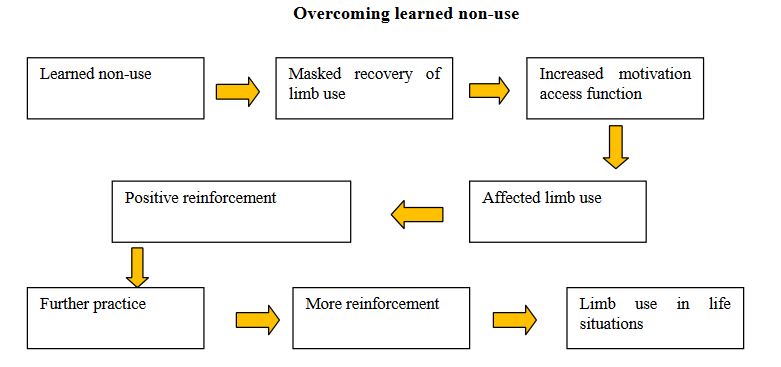
CIMT therapy is based on research by psychologist, Dr. Edward Taub and collaborators at the University of Alabama. Dr. Taub demonstrated that monkeys with a surgical deafferentation (i.e. where somatic sensation was abolished) of a forelimb, ceased using the affected extremity. Through failed attempts to use the deafferented forelimb, the monkeys developed compensation methods to avoid using the affected limb, that is, they effectively learned not to use their affected extremity. Dr. Taub hypothesized that the non-use was a learning mechanism and calls this behavior “Learned non-use”. [4][5]
In the original concept and application in patients, the less affected arm-hand of a patient was immobilized in a sling[6] [7]. This soon progressed to an emphasis on intensive, repetitive training - massed practice - of the more affected arm-hand. In the current application of the method, patients wear a mitt on the less-affected arm 90% of their waking hours and perform repetitive exercises with the more affected arm six to seven hours per day during two to three weeks [8] [9].
Neurophysiological Aspects of CIMT[edit | edit source]
On transcranial magnetic stimulation, an increase in size in the ipsilateral motor cortex region, representing the affected hand muscles of stroke patients has been demonstrated. Furthermore, functional neuroimaging studies (also on stroke patients) have shown varying patterns of change in the activation of the sensorimotor network following CIMT.[10]In a systematic review performed in 2018, studies included also found increased cortical activation and representation as well as significant interhemispheric inhibition of the contralesional hemisphere. These significant neural changes are strongly correlated to improvement in motor function.[11]
CIMT Candidates[edit | edit source]
Diagnoses which may benefit most from CIMT are:
- Stroke (Cerebrovascular accidents)
- Traumatic Brain Injury
- Spinal cord injury
- Multiple sclerosis
- Cerebral palsy (Hemiplegia)
Stroke[edit | edit source]
In a Swedish paper published in 2006, participants who benefitted from CIMT were those who had suffered a stroke and had...[12]
- Some hand function
- High motivation
- Minimal cognitive dysfunction
- Adequate balance
- Adequate walking ability while wearing the restraint to be eligible to participate in CIMT interventions. In a paper published in 2006 by Taub & Uswatte [13], commenting on CIMT on the upper limb for stroke patients, the 10 x 10 x 10 eligibility criteria could be used in selecting a patient for CIMT:
- 10 degrees active wrist extension on the affected hand
- 10 degrees active thumb abduction on the affected hand
- 10 degrees active extension of any other two digits on the affected hand
Also in order for CIMT be most beneficial in the stroke population, it was suggested that these candidates demonstrated the following: [12]
- Limited spasticity (0,1,1⁺) according to modified Ashworth scale.
- Ability to move the affected arm 45 degrees of shoulder flexion and abduction, and 90 degrees of elbow flexion and extension.
- Adequate balance.
- Minimal cognitive dysfunction.
Components of CIMT[edit | edit source]
There are 3 major components of CIMT:[4]
- Shaping
- Task practice
- Packaging of behavioural techniques.
- Shaping is a training method in which a motor task is gradually made more difficult. Shaping programs are individualized, consisting of 10-15 tasks selected primarily from a basic battery of tasks. Each task is usually performed in a set of 10-30 sec trials. At the end of each set of 10 trials, the task is changed. Only one shaping parameter is changed at a time. Shaping requires constant therapist involvement.
- Task practice is repetitive practice of individual functional tasks that takes roughly 15-20mins. Rest is provided as required. Encouragement is given on an infrequent basis (i.e. every 5 mins) with feedback at the end of the task as well about how they performed. Task practice requires less therapist involvement.
- Packaging of behavioural techniques (or the administration of a transfer package) is designed to transfer gains from the clinic to daily life. It includes a behavioral contract that improves adherence to the rehabilitation process. It also includes components such as a log book daily assignments and engaging patients in problem solving. Furthermore, this allows for the identification of barriers and problem-solving to overcome these obstacles. The daily administration of a motor activity log promotes adherence.
Restraints[edit | edit source]
The restraints commonly used for CIMT includes[14]:
- Sing
- Plaster cast
- Triangular bandage
- Splint
- Sling combined with a resting hand splint
- Half glove
- Mitt
Models of CIMT[edit | edit source]
The treatment models are commonly explained in two methods:
1. Unmodified CIMT: Uses a variety of approaches that promote the affected limb for 90% of the individuals waking hours. Only activities involving toileting, hygiene and bathing are permitted. This is done by constraining or reducing the use of the unaffected extremity for 2-3 weeks. The most common form of constraints used for the upper extremity are slings, mitts with velcro or resting hand splints. [15] [16]
2. Modified CIMT: This is more pragmatic model. The program consists of 3 hour per day for 5 days/week, for a minimum of 4 successive weeks. In total there will be 20 treatment sessions totaling to 60 hours. The client is expected to use his/her affected extremity for a minimum of the five “top arm use hours” at home during each week day. [17]
Advantages to CIMT[edit | edit source]
The following have been identified as advantages in the use of CIMT: [18] [19]
- Overall greater improvement in function than traditional treatment.
- Highly researched and highly credible treatment approach.
- There are brain activity and observed gray matter reorganization in primary motor, cortices and hippocampus.
- Increase social participation.
- Decrease in medical cost over lifetime.
Side Effects[edit | edit source]
Few studies have reported harms associated with CIMT, such as burns, minor skin lesions and muscle soreness (stiffness and discomfort) in the affected upper extremity [20] . Shoulder pain in the acute phase after stroke has not been shown to increase after wearing a constraint. Patient endures many hours of frustration.[21]
Evidence[edit | edit source]
The Evidence for Early Intervention[edit | edit source]
Early intervention is important because learning-induced brain plasticity at an early age has a unique impact on brain development. Several researchers have demonstrated that activation and stimulation significantly affect neural activity in the sensory and motor cortex[22]. In young children, there are ongoing structural changes in the corticospinal system directed to hand function [23] . These observed changes are activity dependent[24] . In infants, CIMT can take advantage of the plasticity of the young brain. and recent knowledge of how to provide training with the aim of influencing future development of hand function[25] .
| [26] |
Evidence Based Practice of CIMT[edit | edit source]
Numerous studies show CIMT improves movement on the affected side. CIMT patients showed “large to very large” improvements in the functional use of their affected arm in their daily lives. Scores on a motor activity log (MAL). in which survivors and caregivers noted how well and how much survivors used their impaired arm in daily living improved an average of 1.8 points for those undergoing CIMT. Those in the control group reported no change. In addition, CIMT patients were able to speed their completion of tasks in lab testing while the placebo patients were slower.
Gains in upper extremity function after constraint-induced therapy have been reported in all stages after the onset of stroke[20] .
Chen et al. (2014)[27]: A systematic review and meta-analysis of forty-one RCTs, sixteen reviews, and two clinical guidelines that assessed the effectiveness of constraint induced movement therapy on upper extremity function in children with cerebral palsy.
El-Kafy et al. (2014)[28]: A RCT that examined the effectiveness of a mCIMT protocol in improving upper extremity function in children with congenital hemiplegic cerebral palsy.
Deluca et al. (2006)[29]: A randomized crossover trial of a new form of pediatric rehabilitation was conducted with 18 children with hemiparesis. Pediatric constraint-induced therapy produced significantly greater gains than conventional rehabilitation services.
Zafter et al. (2016)[30]: A RCT that examined the effectiveness of constraint induced movement therapy compared to bimanual therapy in upper motor function in children with hemiplegic cerebral palsy.
Gelkop et al. (2015)[31]: A matched-pair randomized trial that examined the effectiveness of mCIMT and Hand-Arm Bimanual Intensive Training (HABIT) protocols when provided in the school setting.
Home based modified CIMT [32][edit | edit source]
Aim - To compare four weeks of a home-based CIMT program (CMIT home) with a signature two weeks CIMT program (CIMT classic)
Method*n=7 chronic CVA patients (pts) in each group
- CIMT home – Initial one-day training including the instruction of a family member. Then training was performed at the patients home. 2 hrs of daily training at pt’s home with supervision by the instructed family member for 20 consecutive days. The constraint of unaffected hand for 60% of waking hours. Once weekly physio visit pt's home for supervision and adaptation of exercises. Pts received 15 hrs of professional physio supervision in total.
- CMIT classic - Original technique – training administered via physiotherapist 6hrs every weekday for two weeks. Overall 60hrs professional supervision.
- Outcome measures performed prior, immediately after and six months after interventions. Outcomes include Wolf motor function test (WMFT) and Motor activity log (MAL).
Results - Significant improvements in motor function on both WMFT and MAL immediately after and at 6/12 in both groups
Clinical application - A home program may be feasible, effective and requires less therapist supervision, therefore, is cost-effective. Future research requires a larger sample size.
Constraint-Induced Movement Therapy Compared to Dose-Matched Interventions for Upper-Limb Dysfunction in Adult Survivors of Stroke: A Systematic Review with Meta-analysis [33] [edit | edit source]
Aim: To summarize the existing literature examining constraint-induced movement therapy (CIMT), relative to dose-matched control interventions, for upper-limb (UL) dysfunction in adult survivors of stroke.
Methods: CINAHL, Cochrane Library, Embase, NARIC/CIRRIE—Rehabdata, PEDro, PubMed, Scopus, and Web of Science were searched from their inception to February 2011. Trial quality was described using the PEDro scale. The findings were summarized with meta-analysis.
Results: For the 22 trials identified, the mean (SD) PEDro score was 6.4 (1.2). Meta-analysis showed CIMT to be superior to dose-matched interventions based on indicators of UL motor capacity (15 trials, n=432; standardized mean difference [SMD]=0.47, 95% CI, 0.27–0.66) and UL ability (14 trials, n=352; SMD=0.80, 95% CI, 0.57–1.02); Functional Independence Measure scores (6 trials, n=182; mean difference [MD]=5.05, 95% CI, 2.23–7.87); and Motor Activity Log scores (Amount of Use: 12 trials, n=318; MD=1.05, 95% CI, 0.85–1.24; Quality of Movement: 11 trials, n=330; MD=0.89, 95% CI, 0.69–1.08).
Conclusions: Compared to control interventions of equal duration and dose, CIMT produced greater improvements in a variety of indicators of UL function in adult survivors of a stroke with the residual movement of their upper limb.
References[edit | edit source]
- ↑ Fritz SL, Butts RJ, Wolf SL. Constraint-induced movement therapy: from history to plasticity. Expert review of neurotherapeutics. 2012 Feb 1;12(2):191-8.
- ↑ 2.0 2.1 Taub E, Uswatte G. Constraint-induced movement therapy: answers and questions after two decades of research. NeuroRehabilitation 2006; 21(2): 93-95.
- ↑ Barghi A, Mark VW, Taub E. Constraint-Induced Movement Therapy: When Efficacious Motor Therapy Meets Progressive Disease. InNutrition and Lifestyle in Neurological Autoimmune Diseases 2017 Jan 1 (pp. 143-155). Academic Press.
- ↑ 4.0 4.1 Uswatte G, Taub E, Morris D, Barman J, Crago J. Contribution of the shaping and restraint components of Constraint-Induced Movement therapy to treatment outcome. NeuroRehabilitation. 2006;21(2):147-56.
- ↑ Taub E., Uswatte G.(2009). Constraint-induced movement therapy: A paradigm for translating advances in behavioral neuroscience into rehabilitation treatments. In: Berntson G., Cacioppo J., editors.Handbook of neuroscience for the behavioral sciences (Vol. 2, pp. 1296–1319) Hoboken, NJ: Wiley; 2009. (Eds.)
- ↑ Wolf S., Lecraw D., Barton L., Jann B. (1989). Forced use of hemiplegic upper extremities to reverse the effect of learned nonuse among chronic stroke and head-injured patients. Exp. Neurol. 104 125–132
- ↑ Taub E., Miller N. E., Novack T. A., Cook E. W., 3rd, Fleming W. C., Nepomuceno C. S., et al. (1993). Technique to improve chronic motor deficit after stroke. Arch Phys Med Rehabil, 74(4), 347-354.
- ↑ E. Taub, S.L. Wolf. (1997). Constraint induced movement techniques to facilitate upper extremity use in stroke patients. Topics in Stroke Rehabilitation, 3, pp. 38–61
- ↑ Taub E, Uswatte G and Elbert T (2002). New treatments in neuro rehabilitation founded on basic research. Nature Reviews Neuroscience (3) 226-236.
- ↑ Marumoto K, Hosomi M, Koyama T, Furukawa K, Kodama N, Domen K. The neural basis of constraint-induced movement therapy: A diffusion tensor imaging (DTI) study. Neuroscience Research. 2011(71):e251.
- ↑ Abdullahi A. Neurophysiological effects of constraint-induced movement therapy and motor function: A systematic review. International Journal of Therapy and Rehabilitation. 2018 Apr 2;25(4):167-76.
- ↑ 12.0 12.1 Brogårdh C. Constraint Induced Movement Therapy : influence of restraint and type of training on performance and on brain plasticity [Internet] [PhD dissertation]. [Umeå]: Samhällsmedicin och rehabilitering; 2006. (Umeå University medical dissertations). Available from: http://urn.kb.se/resolve?urn=urn:nbn:se:umu:diva-763
- ↑ Taub, E. and Uswatte, G. (2006). Constraint-induced movement therapy: answers and questions after two decades of research. NeuroRehabilitation, 21(2), 93-95.
- ↑ Charles, Jeanne; Gordon, Andrew M. (2005). "A Critical Review of Constraint-Induced Movement Therapy and Forced Use in Children with Hemiplegia". Neural Plasticity. 12 (2–3): 245–61; discussion 263–72.
- ↑ Gauthier LV, Taub E, Mark VW, Perkins C, Uswatte G. Improvement after constraint-induced movement therapy is independent of infarct location in chronic stroke patients. Stroke. 2009 Jul 1;40(7):2468-72.
- ↑ Page S, Levine P, Sisto SA, Bond Q and Johnston MV (2002). Stroke patients´ and therapists´ opinions of constraintinduced movement therapy. Clin Rehabil (16) 55-60.
- ↑ Brogårdh C and Sjölund BH (2006). Constraint induced movement therapy in patients with stroke: a pilot study on effects of small group training and of extended mitt use. Clin Rehabil (20) 218-227.
- ↑ Richards, L., Gonzalez Rothi LJ, Davis S, Wu SS, Nadeau SE.(2006) Limited dose response to Constraint-Induced Movement Therapy in patients with chronic stroke. Clinical Rehabilitation. 20: 1066-1074
- ↑ Sterr, A., Elbert T., Berthold I., Kolbel S and Rockstroh B.(2002). Longer versus shorter daily constraint-induced movement therapy of chronic hemiparesis: and exploratory study. Archives of Physical Medicine & Rehabilitation. 83:1374-1377.
- ↑ 20.0 20.1 Hakkennes S, Keating JL. (2005). Constraint-induced movement therapy following stroke: a systematic review of randomised controlled trials. Aust J Physiother. 51(4):221-31.
- ↑ Ploughman M, Corbett D.(2004). Can forced-use therapy be clinically applied after stroke? An exploratory randomized controlled trial. Arch Phys Med Rehabil. 85(9):1417-23.
- ↑ Nudo, R.J., Milliken GW, Jenkins WM, Merzenich MM.(1996). Use-dependent alterations of movement representations in primary motor cortex of adult squirrel monkeys. Journal of Neuroscience. 16:785-807.
- ↑ Eyre, J.A.,(2007). Corticospinal tract development and its plasticity after perinatal injury. Neurosci Biobehav Rev. 31(8): 1136-49.
- ↑ Martin, J.H., S. Chakrabarty, and K.M. Friel, (2011). Harnessing activity-dependent plasticity to repair the damaged corticospinal tract in an animal model of cerebral palsy. Dev Med Child Neurol. 53 (4):9-13. 4.
- ↑ Friel, K.M., S. Chakrabarty, and J.H. Martin, (2013). Pathophysiological mechanisms of impaired limb use and repair strategies for motor systems after unilateral injury of the developing brain. Dev Med Child Neurol. 55 (4):27-31.
- ↑ physicaltherapyvideo. Stroke Therapy: Constraint Induced Movement Therapy for Arms. Available from: http://www.youtube.com/watch?v=8F-Pgukdjj8 [last accessed 13/08/16]
- ↑ Chen, Y., Pope, S., Tyler, D., & Warren, G. (2014). Effectiveness of constraint-induced movement therapy on upperextremity function in children with cerebral palsy: A systematic review and meta-analysis of randomized controlled trials. Clinical Rehabilitation, 28(10), 939-953.
- ↑ El-Kafy, M. A., Elshemy, S. A., & Alghamdi, M. S. (2014). Effect of constraint-induced therapy on upper limb functions: A randomized control trail. Scandinavian Journal of Occupational Therapy, 21, 11-23.
- ↑ Deluca, S. C.; Echols, K.; Law, C. R.; Ramey, S. L. (2006). "Intensive Pediatric Constraint-Induced Therapy for Children with Cerebral Palsy: Randomized, Controlled, Crossover Trial". Journal of Child Neurology. 21 (11): 931–8.
- ↑ Zafer, H., Amjad, I., Malik, A. N., & Shaukat, E. (2016). Effectiveness of constraint induced movement therapy as compared to bimanual therapy in upper motor function outcome in child with hemiplegic cerebral palsy. Pakistan Journal of Medical Sciences, 32(1), 181-184.
- ↑ Gelkop, N., Burshtein, D. G., Lahav, A., Brezner, A., AL-Oraibi, S., Ferre, C. L., & Gordon, A. M. (2015). Efficacy of constraint-induced movement therapy and bimanual training in children with hemiplegic cerebral palsy in an educational setting. Physical and Occupational Therapy in Pediatrics, 35(1), 24-39.
- ↑ Barzel A, Liepert J, Haevernick K, Eisele M, Ketels G, Rijntjes M, van den Bussche H. Comparison of two types of Constraint-Induced Movement Therapy in chronic stroke patients: A pilot study. Restorative neurology and neuroscience 2009; 27(6): 675-682.
- ↑ Stevenson T, Thalman L, Christie H, Poluha W. Constraint-Induced Movement Therapy Compared to Dose-Matched Interventions for Upper-Limb Dysfunction in Adult Survivors of Stroke: A Systematic Review with Meta-analysis. Physiother Can. 2012;64(4):397-413.