Ehlers-Danlos Syndrome
Original Editors - Corey Vogt from Bellarmine University's Pathophysiology of Complex Patient Problems project.
Top Contributors - Corey Vogt, Eliza Clark, Megan Kanter, Admin, Lucinda hampton, Elaine Lonnemann, Kim Jackson, Laura Ritchie, Abbey Wright, Rosie Swift, 127.0.0.1, Wendy Walker and Blessed Denzel Vhudzijena - Your name will be added here if you are a lead editor on this page.
Introduction[edit | edit source]
Ehlers Danlos syndrome (EDS) is a group of hereditary connective tissue disorders which manifests clinically with skin hyper-elasticity, hypermobility of joints, atrophic scarring, and fragility of blood vessels.
It is largely diagnosed clinically, although identification of the gene encoding the collagen or proteins interacting with it is necessary to identify the type of EDS. It is important to identify the type of EDS to guide management and counselling[1][2]
Epidemiology[edit | edit source]
The incidence of Ehlers-Danlos syndrome, including all subtypes in the general population, is best estimated to be between 1 in 2500 and 1 in 5000[1]
- Hypermobility and classic subtypes are the most common with a prevalence of 1 per 10,000-15,000 and 1 per 20,000-40,000 respectively.
- EDS demonstrates equal prevalence amongst males and females of all racial and ethnic backgrounds. [2][3][4][5][6]
- There are other forms of EDS that are very rare. These include the arthrochalasia, kyphoscoliosis, dermatosparaxis, and vascular types. Currently, only 30 cases of the arthrochalasia and 60 cases of kyphoscoliosis have been reported worldwide.
- The vascular type is the rarest form, affecting about 1 in 250,000 people worldwide. [7]
- While there is no cure for the Ehlers-Danlos syndromes, there is treatment for symptoms, and there are preventative measures that are helpful for most.
Pathophysiology[edit | edit source]
The pathophysiology of most Ehlers Danlos syndrome subtypes involves heritable mutations in collagen synthesis and/or processing[8].
- The inheritance pattern of these mutations is variable, including autosomal dominant and recessive inheritance involving different mutations; there are reports of spontaneous mutations causing identical genotypes and phenotypes.
- The collagen affected by these mutations is integral to every body system, from the skin to the integrity of the vasculature, and as such, the symptoms of the disease can be variable and widespread.
Characteristics/Clinical Presentation[edit | edit source]
Patient presentations vary widely based on the respective underlying subtype.
Ehlers-Danlos Syndrome contains at least six discernible phenotypes that are individually recognised. Each type contain characteristics similar to the others. Each specific type presents with the same general clinical characteristics that are a result of faulty or reduced amounts of Type III collagen in the body:[2][9][4][3][5][6][10]
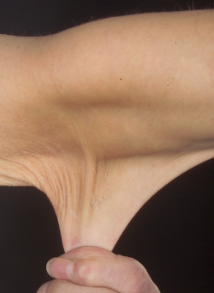
- Cutaneous manifestations are the hallmark of Ehlers-Danlos syndrome: include hyperextensibility, smooth and velvet-like texture, fragility, delayed wound healing, and thin atrophic scars after wound healing.
- Common musculoskeletal manifestations:
- Subluxation
- Dislocations
- Early osteoarthritis
- Persistent pain[11]
- Recurrent fractures
- Hypermobility may manifest as early as peripartum with hip dislocations in the newly delivered infant.
- Other tissues are subject to friability due to the underlying collagen dysfunction:
- Hollow and solid internal organ spontaneous rupture
- Traumatic rupture or perforation
- Hernia
- rectal prolapse
- Neurological manifestations include:
- Hypotonia
- Developmental delay
- Generalized pain[11]
- Insomnia
- Chronic fatigue
- Cardiovascular manifestations include:
- Mitral valve prolapse
- Less commonly, tricuspid valve prolapse.
- Aortic root dilation - can lead to rupture without or without trauma.
- Intracranial arteries, may be aneurysmal, predisposing them to rupture.
- Capillary fragility may manifest as easy bruising and bleeding without underlying bleeding diathesis[1]
Diagnostic Criteria[edit | edit source]
Clinical examination and a detailed family history have proven to be the most effective means of accurately diagnosing EDS.
Major diagnostic criteria typically includes:[2][3][12]
- Joint hypermobility as indicated by a score of greater than or equal to 6/9 on the Beighton scale (Gold standard)
- Soft skin or skin hyperextensibility as defined by >1.5 cm on volar surface of forearm
- Fragile skin or significant skin/soft tissue abnormalities (easy bruising, delayed wound healing, atrophic scarring, easy tendon, ligament, vessel rupture)
For a detailed overview of the diagnostic criteria for specific types of Ehlers Danlos Syndrome,
The definite diagnosis of all Ehlers Danlos Syndrome subtypes, except for except for the hypermobile type, relies on the identification of causative genetic variants[13]. A clinical criteria for hypermobile Ehlers Danlos Syndrome has been developed, in order to allow for a better distinction from other joint hypermobility disorders (see reference[13]).
Systemic Involvement[edit | edit source]
The prognosis depends on the type of EDS and the individual. Life expectancy can be shortened for those with the Vascular Ehlers-Danlos syndrome due to the possibility of organ and vessel rupture. Life expectancy is usually not affected in the other types.[14]
Musculoskeletal
- Joint laxity manifesting as recurrent joint subluxations/dislocations due to minimal trauma and/or spontaneous onset.
- Osteoarthritis resulting in early onset of degenerative joint disease.
- Osteoporosis due to reduction in general bone density up to 0.9 standard deviations lower than the average, healthy adult
- Scoliosis, Kyphosis
- Chronic joint, ligament, tendon, or muscle pain due to myofascial and/or neuropathic source
- Headaches related to muscle tension in cervical spine and TMJ dysfunction
Neuromuscular
- Low muscle tone (hypotonia)
- Generalized muscle weakness
- Decreased reflexes in the knee extensors and flexors seen in adolescents
Neurological
- Fatigue, pain and anxiety are often due to exhaustion of the CNS's reserves
- Migraines often disabling, Chronic pain
- Hyperalgesia is commonly seen in children and adults with hypermobility EDS due their central nervous system being highly sensitized
Cardiopulmonary
- Dysautonomia or Autonomic Dysfunction resulting in abnormal chest pain, palpitations at rest or with exertion, or abnormal blood pressure responses.
- Aortic Root Dilation resulting in predisposition to arterial fragility or rupture. Mitral Valve Prolapse with increased risk of developing infective endocarditis
Gastrointestinal
- Functional Bowel Disorders (gastritis, irritable bowel syndrome, gastroesophageal reflux) occur in up to 50% of individuals with EDS
- High prevalence of GI reflux abdominal pain, constipation and diarrhoea in adolescents with hypermobility EDS
- Hyperextensibility of skin
- Fragility of soft tissue resulting in increased likelihood of rupture or tearing of internal organs
Genitourinary
- Uterine Fragility
- Premature rupture of fetal membranes during pregnancy
- Pelvic prolapse
Oral/Dental
- Periodontal disease resulting in friability, gingivitis, and gum recession, Presence of a high, narrow palate combined with dental crowding
- Easy bruising, Prolonged bleeding times, epistaxis, and menometrorrhagia
Psychiatric
Management[edit | edit source]
Any provider caring for a patient with Ehlers-Danlos syndrome should be aware of the multitude of complications of the disease and potential preventative measures. Treatment and management of patients with EDS should use a multidisciplinary approach that focuses on the prevention of disease progression and subsequent complications as there is no cure for the disease. Specialists generally manage specific care within the field of which the patient has concerning pathology. eg.the monitoring of cardiovascular concerns will be by a cardiologist; likewise, musculoskeletal pathology is monitored and treated by an orthopedist; geneticist or family medicine provider acts as the primary provider referring the patient to these specialists[1]
Treatment of EDS typically consists of management of specific signs and symptoms of the condition as well as lifestyle adjustments to prevent injury/complications. These include:
Education
- Avoidance of high impact activities that place increased stress on pre-morbid lax joints, such as heavy lifting or resistance training
- Avoidance of activities that require joint hyperextension, such as excessive stretching or repetitive activities
- Meticulous skin care
- Meticulous dental care
- Frequent medical check-ups for vascular dysfunction associated with Vascular EDS, bone density (DEXA scans), or orthopaedic dysfunctions associated with increased joint laxity and low muscle tone
Occupational Therapy
| [16] |
- Bracing/splinting in combination with orthopaedists, rheumatologists, and physical therapists to promote increased joint stability and decrease likelihood of joint subluxation/dislocation, especially in upper extremity joints and vertebral joints
Ophthalmologist
- Consultation to screen for myopia, retinal tears, and keratoconus common in individuals with EDS
Dentist
- Consultation to screen for periodontitis and to emphasize importance of meticulous dental care in individuals with EDS
Surgical/Invasive Procedures
Surgical and/or other invasive procedures are not necessarily recommended in patients with EDS as a means of primary treatment due to the impaired wound healing, increased likelihood of scarring, and increased likelihood of blood vessel rupture associated with EDS. However, certain subtypes of EDS, most notably the classic and vascular subtypes of EDS possess an increased predisposition to surgical complications compared to the others.
Physical Therapy Management[edit | edit source]
Physiotherapists play an important role in management through exercise prescription and patient education for many of these conditions.[18].Each physical therapy plan of care must be specially created for the patient depending upon the subtype of EDS and the signs and symptoms of that patient.
In general, physical therapy intervention focuses on decreasing the patient’s disability from a multidirectional approach:
- Aerobic exercise program: consisting of aerobic conditioning combined with a low resistance, high repetition resistive training program to promote increased joint stability by increasing general resting muscle tone
- Strength exercise program: to avoid recurrent joint subluxations/dislocations due to increased muscle tone and to counteract presence of excessive joint, ligament, tendon, and muscle laxity
- Assistive devices: to provide loading relief to lower extremity joints during ambulation and weight bearing activities
- Bracing: to promote increased joint stability and decrease likelihood of joint subluxation/dislocation
- Pain management techniques: to address soft tissue, myofascial, and chronic joint pain associated with EDS
Outcome Measures[edit | edit source]
There is currently not adequate research for specific EDS outcomes.
These are some of the suggested assessment tools used to measure progress of impairments in this population. It is possible many different measurements could be used that focus on balance, gait speed, cadence, dual-task activities, quality of life, and fear of falling.
- Falls Efficacy Scale
- Modified Clinical Test of Sensory Interaction in Balance (mCTSIB)
- Functional Gait Assessment (FGA) / Dynamic Gait Index (DDI)
- Multidimensional Fatigue Symptom Inventory
Differential Diagnosis[edit | edit source]
Include, not limited to:
- Marfans Syndrome
- Loeys-Dietz Syndrome
- Stickler Syndrome
- Williams Syndrome
- Aarskog-Scott Syndrome
- Fragile X Syndrome[3]
EDS is often associated with other persistent pain conditions such as[1]:
Case Study[edit | edit source]
Ehlers-Danlos_Syndrome_Case_Study
Resources[edit | edit source]
Ehlers-Danlos National Foundation
Ehlers-Danlos Syndrome Network C.A.R.E.S
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 1.4 Miklovic T, Sieg VC. Ehlers danlos syndrome. InStatPearls [Internet] 2021 Jul 10. StatPearls Publishing.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Steiner RD. Ehlers-Danlos Syndrome. http://emedicine.medscape.com/article/943567-overview (Accessed Feb 15, 2010).
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 Levy HP. Ehlers-Danlos Syndrome, Hypermobility Type. Gene Reviews. http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=gene&part=eds3 (Accessed Feb 16, 2010).
- ↑ 4.0 4.1 Wenstrup R, Paepe AD. Ehlers-Danlos Syndrome, Classic Type. Gene Reviews. http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=gene&part=eds (Accessed Mar 3, 2010).
- ↑ 5.0 5.1 Yeowell HN, Steinman B. Ehlers-Danlos Syndrome, Kyphoscoliotic Form. Gene Reviews. http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=gene&part=eds6 (Accessed Mar 3, 2010).
- ↑ 6.0 6.1 Pepin MG, Myers PH. Ehlers-Danlos Syndrome, Vascular Type. Gene Reviews. http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=gene&part=eds4 (Accessed Mar 3, 2010).
- ↑ Ehlers-Danlos syndrome. Genetics Home Reference. 2017. Accessed March 2017. Available from: https://ghr.nlm.nih.gov/condition/ehlers-danlos-syndrome#statisticshttps://ghr.nlm.nih.gov/condition/ehlers-danlos-syndrome#statistics
- ↑ Malfait F, Paepe AD. The ehlers-danlos syndrome. Progress in heritable soft connective tissue diseases. 2014:129-43.
- ↑ De Paepe A, Malfait F. The Ehlers–Danlos syndrome, a disorder with many faces. Clinical genetics. 2012 Jul;82(1):1-1.
- ↑ Voermans N, Knoop H, van de Kamp N, Hamel B, Bleijenberg G, van Engelen B. Fatigue Is a Frequent and Clinically Relevant Problem in Ehlers-Danlos Syndrome. 2010;30(3):267-274
- ↑ 11.0 11.1 Rombaut L, Scheper M, De Wandele I, De Vries J, Meeus M, Malfait F, Engelbert R, Calders P. Chronic pain in patients with the hypermobility type of Ehlers–Danlos syndrome: evidence for generalized hyperalgesia. Clinical rheumatology. 2015 Jun;34(6):1121-9.
- ↑ 12.0 12.1 Engelbert RH, Juul‐Kristensen B, Pacey V, De Wandele I, Smeenk S, Woinarosky N, Sabo S, Scheper MC, Russek L, Simmonds JV. The evidence‐based rationale for physical therapy treatment of children, adolescents, and adults diagnosed with joint hypermobility syndrome/hypermobile Ehlers Danlos syndrome. InAmerican Journal of Medical Genetics Part C: Seminars in Medical Genetics 2017 Mar (Vol. 175, No. 1, pp. 158-167).
- ↑ 13.0 13.1 Malfait F, Francomano C, Byers P, Belmont J, Berglund B, Black J, Bloom L, Bowen JM, Brady AF, Burrows NP, Castori M. The 2017 international classification of the Ehlers–Danlos syndromes. InAmerican Journal of Medical Genetics Part C: Seminars in Medical Genetics 2017 Mar (Vol. 175, No. 1, pp. 8-26).
- ↑ EDS society WHAT ARE THE EHLERS-DANLOS SYNDROMES? Available from:https://www.ehlers-danlos.com/what-is-eds/ (accessed 22.2.2021)
- ↑ 15.0 15.1 Castori M, Morlino S, Celletti C, Celli M, Morrone A, Colombi M, Camerota F, Grammatico P. Management of pain and fatigue in the joint hypermobility syndrome (a.k.a. Ehlers–Danlos syndrome, hypermobility type): Principles and proposal for a multidisciplinary approach. Am J Med Genet Part A, 2012;158A(8):2055–2070.
- ↑ Bennett Bremner. Spiral Thigh Brace for Ehlers Danlos Syndrome. Available from: http://www.youtube.com/watch?v=LWNiI-YyfE0 [last accessed 27/09/13]
- ↑ Bathen T, Hångmann AB, Hoff M, Andersen LØ, Rand-Hendriksen S. 2013. Multidisciplinary treatment of disability in Ehlers–Danlos syndrome hypermobility type/hypermobility syndrome: A pilot study using a combination of physical and cognitive-behavioral therapy on 12 women. Am J Med Genet Part A. 2013; 161A(12):3005–3011.
- ↑ 18.0 18.1 Engelbert RH, Juul‐Kristensen B, Pacey V, De Wandele I, Smeenk S, Woinarosky N, Sabo S, Scheper MC, Russek L, Simmonds JV. The evidence‐based rationale for physical therapy treatment of children, adolescents, and adults diagnosed with joint hypermobility syndrome/hypermobile Ehlers Danlos syndrome. InAmerican Journal of Medical Genetics Part C: Seminars in Medical Genetics 2017 Mar (Vol. 175, No. 1, pp. 158-167).
- ↑ Russek LN. Examination and Treatment of a Patient with Hypermobility Syndrome. Physical Therapy 2000;80(4):386-398.
- ↑ Russek LN. Hypermobility Syndrome. Physical Therapy 1999;79(6):591-599.
- ↑ Yeowell HN, Steinmann B. Ehlers-Danlos syndrome, kyphoscoliotic form. NCBI, 2013. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1462/?report=printable
- ↑ Ferrell W, Tennant N, Sturrock R, Ashton L, Creed G, Brydson G et al. Amelioration of symptoms by enhancement of proprioception in patients with joint hypermobility syndrome. Arthritis & Rheumatism. 2004;50(10):3323-3328.