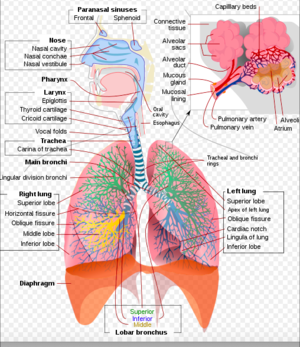
Inhalation Injury
Original Editor - Joseph Ayotunde Aderonmu
Top Contributors - Joseph Ayotunde Aderonmu, Chelsea Mclene, Naomi O'Reilly and Kim Jackson
Introduction[edit | edit source]
Inhalation injury happens to be one of the most challenging injuries for burn care providers. This is because it is one of the classic determinants of mortality that occurs after severe burn injury. The other determinants are age, extent of injury, as well as delay in resuscitation.[1] Inhalation injury refers to pulmonary injury resulting from inhalation of smoke or chemical products of combustion.[2]
Inhalation injury results in direct cellular damage, alterations in regional blood circulation and perfusion, obstruction of the airways, and the release of pro-inflammatory cytokine and toxin release.[3][4] Inhalation injuries also causes reduced functionality of mucociliary clearance and weakening of alveolar macrophages.[5] With this, patient is placed at a high risk of bacterial infection, especially pneumonia, which is one of the top causes of death for burn patients.[6][7]
Epidemiology[edit | edit source]
Inhalation injury is recorded in about one-third of all burn injuries and it is responsible for about 90% of all burn-related mortality.[3][8] The National Burn Repository of the American Burn Association reported up to 10.3% of burn patients to have accompanying inhalation injury.[9] Thus, 1 in 10 burn patients that survive to admission will have the inhalation injury and a corresponding increase in the mortality rate.[10]
Classes[edit | edit source]
Anatomically, inhalation injuries are divided into three classes:[11]
Heat Injury to the Upper Airway[edit | edit source]
When the room air temperature reaches 1000°F after a fire outbreak, injury results to the structures of the airway above the carina. This is due to the combination of efficient heat dissipation in the upper airway, low heat capacity of air and reflex closure of the larynx.[12]
The result of the injury to these airway structures include extensive swelling of the the tongue, epiglottis, and aryepiglottic folds and accompanying obstruction. It takes a period of hours for airway swelling to develop as fluid resuscitation is taking place. It is important to note that initial evaluation might not be the best indicator of the extent of the obstruction that may later occur.[13]
Chemical Injury to the Lower Airways[edit | edit source]
Combustion of materials leads to the the production of toxic materials to the respiratory tract. This may cause local chemical irritation in the respiratory tract.[2] Sulfur dioxide is produced by burning rubber and plastic, as well as other gases such as nitrogen dioxide, ammonia and chlorine with strong acids and alkali after combination with water in the respiratory airways and alveoli. Also, laminated furniture may contain glues that may release cyanide gas during combustion. Aldehydes are also produced when cotton or wool are burned. Furthermore, toxins produced by smoke may damage airway epithelial and capillary endothelial cells.[11][14]
Systemic Toxicity due to Carbon Monoxide or Cyanide Exposure[edit | edit source]
Fatalities result from carbon monoxide poisoning in burn-injured patients. Unfortunately, many of its fatalities happen at the scene of the fire due to its mechanism. In a closed fire space, injury is influenced by the levels of carboxyhemoglobin. Significant injury can result in just a short time frame with the exposure to carboxyhemoglobin levels as minute as 10%.[11][15][16] Carbon monoxide is a competitive inhibitor of intracellular cytochrome oxidase enzyme systems, especially the cytochrome P-450 resulting in inactivation of cellular systems to make use of oxygen.[2]
On the other hand, inhaled hydrogen cyanide, which is a product of the combustion of multiple household materials also inhibits the cytochrome oxidase system. This then foster a synergy with carbon monoxide to cause tissue hypoxia with acidosis and a reduction in the consumption of oxygen by the brain tissues.[11][16]
Pathophysiology[edit | edit source]
What determines the amount of damage caused by an inhalation injury, are the environment and the host. Also, other factors such as the injury source, the gases produced (temperature, concentration, and solubility), and the response of an individual to the injury determine the amount of damage.[17] The effects that follow inhalation injuries include formation of casts, reduction in the amount of available surfactants, increased airway resistance, and reduction in pulmonary compliance.[18] These culminate in acute lung injury and acute respiratory distress syndrome.[19]
This mechanism of destruction in inhalation injuries can be classified in one of four ways:
Upper Airway Injury[edit | edit source]
The course of this pathophysiology is induced by microvascular changes that result after direct thermal injury and chemical irritation.[6] Heat produced from the burn denatures protein. This causes the activation of the complement cascade that results in the release of histamine.[17][20] Furthermore, xanthine oxidase is formed, reactive oxygen species (ROS) are released which reacts with nitric oxide in the endothelium to cause upper airway edema by raising the pressure of the microvasculature and local permeability.[17][21][22] Also, pro-inflammatory cytokines, with ROS and eicosanoids bring polymorphonuclear cells to this area further causing the release of ROS and signaling proteases.[1]
This causes a remarkable increase in the pressure of the microvasculature, a reduction in interstitial hydrostatic pressure and an increase in interstitial oncotic pressure.[17] Since burn patients are administered crystalloids to be used in resuscitation, this further reduces the plasma oncotic pressure, affecting the oncotic pressure gradient in the microcirculation causing more significant airway edema.[17] Without steam inhalation and blast injuries, an efficient protection is given to the lower airway by the upper airway through heat exchange to restrict distal damage to the lower airways.[1]
Lower Airway Injury[edit | edit source]
Lower airway injury occurs as a result of the chemicals in smoke. Due to the low heat capacity of air and the efficient bronchial circulation regulating the temperature of the airway gases, most gases are at body temperature when the pass through the glottis.[23] For the induction of thermal injury to the airways, flames have to be in direct contact.[24] Burned biological materials are toxic to the airways and cause an initial response to trigger proinflammatory response. This causes up to a 10-fold increase in bronchial blood circulation within minutes after an inhalation injury. [16] This increase is sustained and results in increased permeability and damage of the bronchial epithelium.[17] An increase in pulmonary transvascular fluid results and a fall in PaO2/FiO2 ≤ 200 within 24 hours following the injury.[25] Furthermore, hyperemia of the tracheobronchial tree and lower airways occurs, which is a clinical finding very common in inhalation injury and is used to often diagnose the injury.[26][27] The copious and foamy secretions that are formed from goblet cells later solidify, resulting in the formation of casts and airway obstruction.[17]
Pulmonary Parenchymal Injury[edit | edit source]
The changes to lung parenchyma occur much later after the injury. The extent of this changes are dependent on the extent of the injury and the response of the patient to the injury. The occurrence of parenchymal injuries are associated with an elevation of pulmonary transvascular fluid levels and this is proportional to the period of exposure to toxins and smokes.[2] Again, it is rare for injury to the lower airways and lung parenchyma to be caused by direct thermal contact. Thus, it is only steam that can overcome the efficient heat dissipating system of the upper airway. There is a decrease to the permeability of protein, an elevation to the permeability to small particles, a reduction in pressure in the pulmonary microvasculature pressure, and hypoxic pulmonary vasoconstriction loss.[17] The major derangements that follow inhalation injury include edema, lowered pulmonary compliance following extravascular lung water and pulmonary lymph, and sudden inactivation of surfactant. In addition, a subsequent ventilation-perfusion mismatch can also occur that can lead to profound hypoxemia and ARDS.[6]
Systemic Toxicity[edit | edit source]
Inhalation of chemicals, cytotoxic liquids, fumes, mist and gases can cause systemic toxic changes. Smoke can combine with these toxins and cause increased mortality by promoting tissue hypoxia, metabolic acidosis, and reducing cerebral oxygen consumption and metabolism.[16][28]
Diagnosis[edit | edit source]
Initially, the diagnosis of inhalation injury was based on the following indirect observations:[29]
- Facial burns
- Singed nasal vibrissae
- A history indicating a burn injury that occurred in an enclosed space
For each of these signs, there is a high level of false positivity. Also, when they are taken together, they are shown to underestimate the true occurrence of inhalation injury. A classic sign of smoke inhalation is also carbonaceous secretions. Although, it is a less exact predictor of either the presence or severity of inhalation injury than is widely believed. Yet, a telltale of exposure to smoke could be carbonaceous secretions but should not confirm either the diagnosis of inhalation injury or its sequela. Hypoxia, rales, rhonchi and wheezes are not often present on admission. But when they occur, they are seen in patients with the most severe injury and this may mean an extremely poor prognosis.[6]
Also, the chest X-ray of patient on admission has also been shown to be a poor indicator of inhalation injury.[6] Yet, it is observed that about two-thirds of patients may have changes of diffuse or focal infiltrates or pulmonary edema between five to ten days after injury. Therefore, the admission film is usually not used for diagnostic purposes, but is useful for making baseline evaluations.[30]
Currently, the standard for the diagnosis of inhalation injury is fiberoptic bronchoscopy.[31] It is however useful only in detecting upper airway injury. The observations may include presence of soot, mucosal necrosis, char, edema of the respiratory airways and inflammation.[32]
However, bronchoscopy alone cannot rule out the possibility of parenchyma damage. Therefore, to find out parenchyma damage, Xenon scanning has been generally utilized.[33] It is a safe, quick test which requires a minimum of patient's cooperation. It involves several chest scintiphotograms once an initial radioactive Xenon gas has been intravenously injected. The test demonstrates the locations of the decreased alveolar gas washout, revealing the sites of tiny airway obstruction that results from edema or fibrin cast formation.[6]
Management[edit | edit source]
There is generally no single standard management protocol for inhalation injury.[1] The mainstay of treatment of inhalation injury is supportive care. This is achieved by acute hospitalization and rehabilitation. [1]
Supportive Care[edit | edit source]
Inhalation injuries result in the formation of casts, they decrease the amount of surfactant available, they elevate airway resistance, and they reduce pulmonary compliance.[18]
Bronchodilators[edit | edit source]
Bronchodilators decrease airflow resistance and improve airway compliance. Albuterol and salbutamol which are β2-adrenergic agonists reduce airway pressure by causing a relaxation of smooth muscles and inhibiting bronchospasm to increase the PaO2/FiO2 ratio.[34]
Muscarinic Receptor Antagonists[edit | edit source]
To reduce airway pressures and mucus secretion, muscarinic receptor antagonists like tiotropium are employed to limit cytokine release through smooth muscle constriction in the airways, and to cause a stimulation of submucosal glands.[35][36]
To reduce the inflammatory response of the host after an inhalation injury, muscarinic receptor antagonists and beta agonists can be used. Structurally, muscarinic and adrenergic receptors are in the lining of the respiratory tract, although its impact on the inflammatory and host response is not fully understood. They however have been demonstrated to reduce the activity of pro-inflammatory cytokines following stress.[37]
Inhaled (Nebulized) Mucolytic Agents and Anticoagulants[edit | edit source]
To address the airway obstruction that occurs following mucus, fibrin cast formation and cellular debris after an inhalation injury, mucolytic agents are used; in particular, N Acetylcysteine (NAC).[38] NAC has anti-inflammatory properties and it is an antioxidant and free radical scavenger.[39] It acts as a strong mucolytic agent that reduces the damage caused by ROS. Inhaled anticoagulants are also employed to reduce airway obstruction from fibrin casts.
Respiratory Support[edit | edit source]
Since a significant upper airway edema usually results from an inhalation injury, and the resuscitation of the burn injury often worsen the airway edema, it is often important to obtain and maintain a patent airway in the management of inhalation injury.[6] [1]
Limited trials have been carried out on the respiratory modes suitable for patients with inhalation injury. Yet, a mechanical ventilation strategy that has been demonstrated to improve morbidity and mortality from acute respiratory distress symptom and acute lung injury comes from the ARDSNET trial. This trial revealed from a large randomized controlled trial that "lung protective strategies of limited tidal volumes of 6–8mL/kg and plateau pressures of less than 30cm H2O improved outcomes".[40]
Since conventional mechanical ventilation modes such as control mode ventilation, assist-control mode, synchronized intermittent mandatory ventilation, pressure control mode and pressure support mode have been limitedly studied in the patient with inhalation injury,[18][41] non-conventional modes of ventilation modes are often employed in order to support patients with inhalation injury and apply lung-protective ventilation strategies.[42]
Common non-conventional ventilator modes that are usually used include high-frequency percussive ventilation (HFPV), high frequency oscillatory ventilation (HFOV), airway pressure release ventilation (APRV), extracorporeal membrane oxygenation (ECMO). Although HFPV has been shown to be the most promising among these modes.[1]
Physiotherapy[edit | edit source]
A number of studies have demonstrated that a techniques such as gravity-assisted bronchial drainage when combined with chest percussion and vibrations. are effective in the removal of secretions .[43][44][45]
Bronchial Drainage / Positioning[edit | edit source]
This is a modality that employs the use of gravity-assisted positioning targeted at improving the hygiene of pulmonary system in patients with inhalation injury and/or retained secretions. Due to skin grafts, donor sites, and the use of air fluid beds, clinical judgment might influence the most appropriate decisions.[6] In fact, positioning in the Trendelenburg and various other positions may acutely worsen hypoxemia. It has been shown that a patient may experience a decline in the level of arterial oxygenation positioning.[45]
Percussion[edit | edit source]
Percussion allows secretions to be removed from the tracheobronchial tree. It is essential to position a suitable padding between the patient and the physiotherapist's hands to prevent skin irritation during the process of percussion.[6] Percussion is applied over the bronchial segments to be drained using their surface landmarks. Incisions, skin grafts, and bony prominence should be avoided during percussion.[46]
Vibration / Shaking[edit | edit source]
Vibration / Shaking mobilizes loosened secretions into larger airways to enable easy cough up or removal through suctioning. Vibrations can be performed mechanically, and this type of vibrations have also been reported to produce good clinical results. For patients who cannot tolerate manual percussion, gentle mechanical vibration may be indicated.[6]
Early Ambulation[edit | edit source]
To prevent respiratory complications, ambulation can be commenced early for patients with inhalation injury. Patients who are on continuous ventilatory support can also be placed into a chair with appropriate use of analgesics. There are establish therapeutic effects of the sitting position which include:[6]
- The patient can breathe with lungs regions that are normally hyperventilated
- Muscular strength and tone can be preserved
- Contractures are prevented and exercise tolerance is maintained
Prognosis[edit | edit source]
It is noteworthy that mortality rates for inhalation injury have not changed over the past five decades, though improvements in standards of care for severe burn injuries have.[1] Supportive strategies are vital in the management of inhalation injury. Yet, more trials are needed to demonstrate sufficient evidence for many of the pharmacological agents. Also, more promising results have been achieved with unconventional modes of ventilation such as HFPV in addressing physiologic derangements from inhalation injury.[1]
Conclusion[edit | edit source]
Inhalation injury requires a robust knowledge of its pathophysiology to guide accurate diagnosis and drive the right therapeutic strategies. Practitioners must carefully work within available evidence for best outcomes from inhalation injuries – a classic determinant of mortality in severe burn.
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Jones SW, Williams FN, Cairns BA, Cartotto R. Inhalation injury: pathophysiology, diagnosis, and treatment. Clinics in plastic surgery. 2017 Jul 1;44(3):505-11.
- ↑ 2.0 2.1 2.2 2.3 Dries and EndorfScandinavian Journal of Trauma, Resuscitation and Emergency Medicine2013,21:31
- ↑ 3.0 3.1 Kadri SS, Miller AC, Hohmann S, Bonne S, Nielsen C, Wells C, Gruver C, Quraishi SA, Sun J, Cai R, Morris PE. Risk factors for in-hospital mortality in smoke inhalation-associated acute lung injury: data from 68 United States hospitals. Chest. 2016 Dec 1;150(6):1260-8.
- ↑ Reper P, Heijmans W. High-frequency percussive ventilation and initial biomarker levels of lung injury in patients with minor burns after smoke inhalation injury. Burns. 2015; 41:65–70. [PubMed: 24986596]
- ↑ Al Ashry HS, Mansour G, Kalil AC, Walters RW, Vivekanandan R. Incidence of ventilator associated pneumonia in burn patients with inhalation injury treated with high frequency percussive ventilation versus volume control ventilation: A systematic review. Burns. 2016 Sep 1;42(6):1193-200.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 Mlcak RP, Suman OE, Herndon DN. Respiratory management of inhalation injury. burns. 2007 Feb 1;33(1):2-13.
- ↑ Pruitt BA, McManus AT. The changing epidemiology of infection in burn patients. World journal of surgery. 1992 Jan 1;16(1):57-67.
- ↑ Tan A, Smailes S, Friebel T, Magdum A, Frew Q, El-Muttardi N, Dziewulski P. Smoke inhalation increases intensive care requirements and morbidity in paediatric burns. Burns. 2016 Aug 1;42(5):1111-5.
- ↑ Burn Incidence Fact Sheet. American Burn Association; 2016 National Burn Repository. American Burn Association; 2017.
- ↑ Foncerrada G, Culnan DM, Capek KD, González-Trejo S, Cambiaso-Daniel J, Woodson LC, Herndon DN, Finnerty CC, Lee JO. Inhalation injury in the burned patient. Annals of plastic surgery. 2018 Mar;80(3 Suppl 2):S98.
- ↑ 11.0 11.1 11.2 11.3 McCall JE, Cahill TJ. Respiratory care of the burn patient. Journal of Burn Care & Rehabilitation. 2005 May 1;26(3):200-6.
- ↑ Pruitt Jr BA, Flemma RJ, DiVincenti FC, Foley FD, Mason Jr AD, Young Jr WG. Pulmonary complications in burn patients: a comparative study of 697 patients. The Journal of thoracic and cardiovascular surgery. 1970 Jan 1;59(1):7-20.
- ↑ Palmieri TL. Inhalation injury: research progress and needs. Journal of burn care & research. 2007 Jul 1;28(4):549-54.
- ↑ Trunkey DD:Inhalation injury. Surg Clin North Am1978,58:1133–1140
- ↑ Moore SJ, Ho K, Hume AS. Severe hypoxia produced by concomitant intoxication with sublethal doses of carbon monoxide and cyanide. Toxicology and applied pharmacology. 1991 Jul 1;109(3):412-20.
- ↑ 16.0 16.1 16.2 16.3 Traber, D., Herndon, David, Enkhbaatar, Perenlei, Maybauer, Marc, Maybauer, Dirk. The pathophysiology of inhalation injury. In: Herndon, D., editor. Total Burn Care. Fourth. Saunders Elsevier; 2012. p. 219-28
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 17.6 17.7 Sousse LE, Herndon DN, Andersen CR, Ali A, Benjamin NC, Granchi T, Suman OE, Mlcak RP. High tidal volume decreases adult respiratory distress syndrome, atelectasis, and ventilator days compared with low tidal volume in pediatric burned patients with inhalation injury. Journal of the American College of Surgeons. 2015 Apr 1;220(4):570-8.
- ↑ 18.0 18.1 18.2 Jones, SW., Ortiz-Pujols, Shiara M., Cairns, Bruce. Smoke inhalation injury: a review of the pathophysiology, management, and challenges of burn-associated inhalation injury. In: Gilchrist ICaEYC. , editor. Current Concepts in Adult Critical Care Society of Critical Care Medicine. Vol. 2011. 2011.
- ↑ Friedl HP, Till GO, Trentz O, Ward PA. Roles of histamine, complement and xanthine oxidase in thermal injury of skin. The American journal of pathology. 1989 Jul;135(1):203.
- ↑ Granger DN. Role of xanthine oxidase and granulocytes in ischemia-reperfusion injury. American Journal of Physiology-Heart and Circulatory Physiology. 1988 Dec 1;255(6):H1269-75.
- ↑ Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox biology. 2015 Dec 1;6:524-51.
- ↑ Baile EM, Dahlby RW, Wiggs BR, Pare PD. Role of tracheal and bronchial circulation in respiratory heat exchange. Journal of Applied Physiology. 1985 Jan 1;58(1):217-22.
- ↑ Moritz AR, Henriques Jr FC, McLean R. The effects of inhaled heat on the air passages and lungs: an experimental investigation. The American journal of pathology. 1945 Mar;21(2):311.
- ↑ Herndon DN, Barrow RE, Traber DL, Rutan TC, Rutan RL, Abston S. Extravascular lung water changes following smoke inhalation and massive burn injury. Surgery. 1987 Aug 1;102(2):341-9.
- ↑ Walker PF, Buehner MF, Wood LA, Boyer NL, Driscoll IR, Lundy JB, Cancio LC, Chung KK. Diagnosis and management of inhalation injury: an updated review. Critical Care. 2015 Dec;19(1):1-2.
- ↑ You K, Yang HT, Kym D, Yoon J, Cho YS, Hur J, Chun W, Kim JH. Inhalation injury in burn patients: establishing the link between diagnosis and prognosis. Burns. 2014 Dec 1;40(8):1470-5.
- ↑ Pitt BR, Radford EP, Gurtner GH, Traystman RJ. Interaction of carbon monoxide and cyanide on cerebral circulation and metabolism. Archives of Environmental Health: An International Journal. 1979 Sep 1;34(5):354-9.
- ↑ Moylan JA, Chan CK. Inhalation injury--an increasing problem. Annals of surgery. 1978 Jul;188(1):34.
- ↑ Putman CE, Loke JA, Matthay RA, Ravin CE. Radiographic manifestations of acute smoke inhalation. American Journal of Roentgenology. 1977 Nov 1;129(5):865-70.
- ↑ Wanner A, Cutchavaree A. Early recognition of upper airway obstruction following smoke inhalation. American Review of Respiratory Disease. 1973 Dec;108(6):1421-3.
- ↑ Moylan JA, Adib K, Birnbaum M. Fiberoptic bronchoscopy following thermal injury. Plastic and Reconstructive Surgery. 1975 Nov 1;56(5):598.
- ↑ Moylan Jr JA, Wilmore DW, Mouton DE, Pruitt Jr BA. Early diagnosis of inhalation injury using 133 xenon lung scan. Annals of surgery. 1972 Oct;176(4):477.
- ↑ Lange M, Hamahata A, Traber DL, Cox RA, Kulp GA, Nakano Y, Traber LD, Herndon DN, Enkhbaatar P. Preclinical evaluation of epinephrine nebulization to reduce airway hyperemia and improve oxygenation after smoke inhalation injury. Critical care medicine. 2011 Apr;39(4).
- ↑ Dries DJ, Endorf FW. Inhalation injury: epidemiology, pathology, treatment strategies. Scandinavian journal of trauma, resuscitation and emergency medicine. 2013 Dec 1;21(1):31.
- ↑ Jonkam C, Zhu Y, Jacob S, Rehberg S, Kraft E, Hamahata A, Nakano Y, Traber LD, Herndon DN, Traber DL, Hawkins HK. Muscarinic receptor antagonist therapy improves acute pulmonary dysfunction after smoke inhalation injury in sheep. Critical care medicine. 2010 Dec 1;38(12):2339-44.
- ↑ van der Poll T, Coyle SM, Barbosa K, Braxton CC, Lowry SF. Epinephrine inhibits tumor necrosis factor-alpha and potentiates interleukin 10 production during human endotoxemia. The Journal of clinical investigation. 1996 Feb 1;97(3):713-9.
- ↑ Suter PM, Domenighetti G, Schaller MD, Laverrière MC, Ritz R, Perret C. N-acetylcysteine enhances recovery from acute lung injury in man: a randomized, double-blind, placebo-controlled clinical study. Chest. 1994 Jan 1;105(1):190-4.
- ↑ Villegas L, Stidham T, Nozik-Grayck E. Oxidative stress and therapeutic development in lung diseases. Journal of pulmonary & respiratory medicine. 2014 Aug;4(4).
- ↑ Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. New England Journal of Medicine. 2000 May 4;342(18):1301-8.
- ↑ Petrucci N, Iacovelli W. Lung protective ventilation strategy for the acute respiratory distress syndrome. Cochrane Database of Systematic Reviews. 2007(3).
- ↑ Fitzpatrick JC, Cioffi Jr WG. Ventilatory support following burns and smoke-inhalation injury. Respiratory care clinics of North America. 1997 Mar 1;3(1):21-49.
- ↑ Chopra SK, Taplin GV, Simmons DH, Robinson Jr GD, Elam D, Coulson A. Effects of hydration and physical therapy on tracheal transport velocity. American Review of Respiratory Disease. 1977 Jun;115(6):1009-14.
- ↑ Marini JJ, Pierson DJ, Hudson LD. Acute lobar atelectasis: a prospective comparison of fiberoptic bronchoscopy and respiratory therapy. American Review of Respiratory Disease. 1979 Jun;119(6):971-8.
- ↑ Oldenburg Jr FA, Dolovich MB, Montgomery JM, Newhouse MT. Effects of postural drainage, exercise, and cough on mucus clearance in chronic bronchitis. American Review of Respiratory Disease. 1979 Oct;120(4):739-45.
- ↑ 45.0 45.1 Remolina C, Khan AU, Santiago TV, Edelman NH. Positional Hypoxemia in Unilateral Lung Disease. Survey of Anesthesiology. 1982 Feb 1;26(1):1.
- ↑ Herndon DN, Inhalation injury. In: Total burn care; 2nd ed., 2002, p. 242–53.