Multiple System Atrophy
Original Editors - Emily Nicklies from Bellarmine University's Pathophysiology of Complex Patient Problems project.
Top Contributors - Emily Nicklies, Lucinda hampton, Admin, Kim Jackson, Dave Pariser, Wendy Walker, Elaine Lonnemann, Naomi O'Reilly, WikiSysop and Amrita Patro
Introduction[edit | edit source]
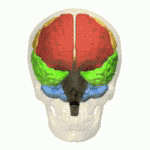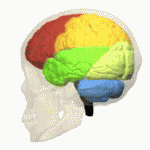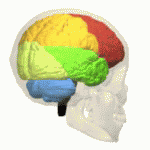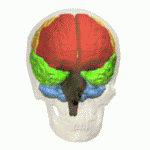
Multiple System Atrophy (MSA) is defined as a sporadic, fatal, progressive, neurodegenerative (specifically a synucleinopathy) adult-onset disorder. It is the second most common neurodegenerative movement disorder with an average annual incidence rate of 3 per 100000 person-years after Parkinson’s disease (PD)[1]. It can affect the
- Autonomic system causing autonomic failure, causing eg.fainting spells and problems with heart rate, erectile dysfunction, and bladder control.
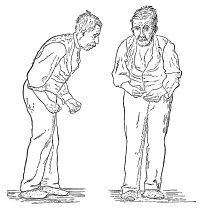
- Basal ganglia causing parkinsonism, causing eg tremor, rigidity, and/or loss of muscle coordination as well as difficulties with speech and gait.
- Cerebellum causing ataxia [2]
The symptoms reflect the progressive loss of function and death of different types of nerve cells in the brain and spinal cord.
Some of these features are similar to those seen in Parkinson’s disease, and early in the disease course it often may be difficult to distinguish these disorders. This 1 minute video is titled Differentiating Multiple System Atrophy From Parkinson's Disease.
Etiology[edit | edit source]
The causes of MSA are unknown; however, as for other neurodegenerative diseases, a complex interaction of genetic and environmental mechanisms seems likely.[4]
Research has suggested that MSA is characterized by a progressive loss of neuronal and oligodendroglial cells (myelin producing cells in CNS) in numerous sites in the CNS (basal ganglia, cerebellum). This damage is said to occur from the formation of glial cytoplasmic inclusions (GCI’s).[5]
Classification[edit | edit source]
MSA is classified by the most dominant symptom exhibited by the patient.
- Parkinsonian type (MSA-P): this includes symptoms similar to Parkinson's disease eg tremors at rest, rigidity of muscles, and slow movements, including gait
- Cerebellar type (MSA-C): involves difficulty walking (ataxia), issues maintaining balance, and trouble coordinating voluntary movements[6].
Epidemiology[edit | edit source]
MSA is an orphan disease with an incidence of up to 2.4 cases per 100 000 persons per year, while the prevalence may reach up to 7.8 patients per 100 000 population over the age of 40. The MSA‐P variant seems to be more common in the western hemisphere, whereas MSA‐C appears to be more frequent in Asia[7] .
There is about 1 living case of MSA in the population for every 40 cases of Parkinson’s disease, but because survival in MSA is shorter than for PD, about 1 new MSA case presents every year for about every 20 who present with PD.[7]
Symptoms[edit | edit source]
The symptoms reflect the progressive loss of function and death of different types of nerve cells in the brain and spinal cord. The initial symptoms of MSA are often difficult to distinguish from the initial symptoms of Parkinson’s disease and include: slowness of movement, tremor, or rigidity (stiffness); clumsiness or incoordination; impaired speech, a croaky, quivering voice; fainting or lightheadedness due to orthostatic hypotension; bladder control problems, eg a sudden urge to urinate or difficulty emptying the bladder[7].
The 2 minute video below gives a good introduction to MSA
Cognition[edit | edit source]
Cognitive deficits occur in all cognitive domains. The impaired cognitive domains had a significant correlation with atrophy of frontotemporal cortical areas, basal ganglia, thalamus, and cerebellum[1]. Cognitive impairment, particularly seen in executive function, may occur in up to 75% of patients The Mini-Mental State Examination, or MMSE, yielded 3% of participants showing signs of cognitive decline while a test called the Frontal Assessment Battery (FAB) categorized 41% of participants as being cognitively impaired. The FAB is a cognitive test that incorporates several clinical assessments to screen for frontotemporal dementia (FTD)
- Emotional instability may occur later in the progression of the disease
- Depression, anxiety, panic attacks, and suicidal ideation may present in MSA
- Patients with MSA can have great difficulty switching attention from one stimulus to another,[9] with a decrease in goal-oriented cognitive ability.
Diagnosis[edit | edit source]
At this time, there are no specific symptoms, blood tests or imaging studies that distinguish MSA[10]. It can take a while to receive an MSA diagnosis because of the similarities between MSA and other conditions, eg Parkinson's disease. MSA is usually diagnosed around 50 years of age and is seen in people of all ethnic backgrounds. Once symptoms begin, the disease tends to progress quite rapidly.
The Mini-Mental State Examination, or MMSE, yielded 3% of participants showing signs of cognitive decline while a test called the Frontal Assessment Battery (FAB) categorized 41% of participants as being cognitively impaired[11]. The FAB is a cognitive test that incorporates several clinical assessments to screen for frontotemporal dementia (FTD),
Diagnostic Challenge There is no “typical” presentation for this disorder, and MSA can often be masked by other diagnoses. Patients with MSA are most commonly misdiagnosed with Parkinson’s Disease. About 1/3 of people with MSA die while still misdiagnosed.[2] Only 25% of patients with MSA are correctly diagnosed at their first neurological visit. The correct diagnosis is usually established on an average of 4 to 5 years after the disease onset.[12]
Management[edit | edit source]
Currently, there is no cure for MSA, nor are there any treatments specifically designed to reverse or stop disease progression.
- Movement disorders can be treated with levodopa and carbidopa (Sinemet), but this usually has limited results.
- Other medications such as trihexyphenidyl (Artane), benztropine mesylate (Cogentin), and amantadine (Symmetrel), may also offer some symptom relief. Several medications exist to treat orthostatic hypertension.
- Physical and occupational therapy, eg aqua therapy, can help maintain muscle function, and speech therapy can help improve any difficulties swallowing or speaking[6].
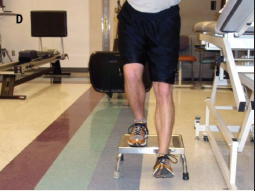
Physical Therapy Management[edit | edit source]
Physical therapies offer drug-free tools to maintain their function for as long as possible, by helping keep muscles strong and flexible and helping prevent falls. Encouraging mobility also lowers the risk of pulmonary embolism which can be fatal[10].
Ideally a neurological physiotherapist will assess the client and then make a tailored program. It incorporates a combination of progressive fitness, balance, movement and strength training and stretching, as well as education on how to manage symptoms. Make PT goals realistic. Base the goals upon current function and take into consideration the activities most important to the patient.
Gait/Mobility Training:
- Fit the patient for the most appropriate walking aids in order to gain independent mobility. This will depend upon the patient’s level of function and safety.
- If a wheelchair is most appropriate, ensure the chair is one that will best benefit their posture and mobility based upon the function of patient. As a PT, the need to promote independent functional mobility and optimal posture with safety in mind is most important.
Therapeutic Exercise/Activities:
- Transfer training
- Balance activities[13]eg Otago balance porogram
- Strength training focusing on knee extensors and flexors, hip abductors and adductors, and ankle plantar flexors .These muscle groups are chosen because of their importance to balance.[13] Can lead to improvements in the patient's gait pattern (better at maintaining good step height and length even) and reduces festinating gait.
- Postural muscle strengthening to improve eg head and trunk alignment in both sitting and standing).[13]
Education
- Education of the patient and family on benefits of PT is important for patient/family knowledge and compliance.
Differential Diagnosis[edit | edit source]
MSA often presents as the following disorders: Pure Parkinsonism[14]; Pure Autonomic Failure (PAF)[14]; Progressive Supranuclear Palsy[15]; Corticobasal Ganglionic Degeneration[16]
Prognosis and Outlook[edit | edit source]
Prognosis is currently guarded, with most MSA patients passing away from the disease or its complications within 6-10 years after the onset of symptoms. Nonetheless, there is reason for hope for MSA research goes on. Since the biology of MSA may be related to other neurodegenerative diseases like Parkinson's disease, it is possible that therapies designed for other conditions will also prove helpful for patients with MSA.[10]
References[edit | edit source]
- ↑ 1.0 1.1 Dash S, Mahale R, Netravathi M, Kamble NL, Holla V, Yadav R, Pal PK. Cognition in Patients With Multiple System Atrophy (MSA) and Its Neuroimaging Correlation: A Prospective Case-Control Study. Cureus. 2022 Jan 29;14(1). Available:https://www.cureus.com/articles/83909-cognition-in-patients-with-multiple-system-atrophy-msa-and-its-neuroimaging-correlation-a-prospective-case-control-study (accessed 14.3.2022)
- ↑ 2.0 2.1 Lundy-Eckman, L. Neuroscience: Fundamentals for Rehabilitation. 3rd ed. St. Louis: Saunders Elsevier, 2002.
- ↑ MSA coalition Differentiating Multiple System Atrophy From Parkinson's Disease (PSA) Available: https://www.youtube.com/watch?v=CD7UL12GSCM(accessed 14.3.2022)
- ↑ Jellinger KA. Multiple system atrophy: an oligodendroglioneural synucleinopathy. Journal of Alzheimer's Disease. 2018 Jan 1;62(3):1141-79 Available from.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5870010/ (last accessed 16.1.2020)
- ↑ Diedrich A, Robertson D. Multiple System Atrophy. Vanderbilt University School of Medicine 2009. http://emedicine.medscape.com/article/1154583-overview (accessed on 24 Jan 2010).
- ↑ 6.0 6.1 Very well health MSA Available: https://www.verywellhealth.com/multiple-system-atrophy-2860862(accessed 14.3.2022)
- ↑ 7.0 7.1 7.2 Virtual strategy Multiple System Atrophy Epidemiology Forecast to 2030 Available: https://virtual-strategy.com/2020/07/16/multiple-system-atrophy-epidemiology-forecast-to-2030/ (accessed 14.3.2022)
- ↑ MSA coalition. What is MSA available from:https://www.youtube.com/watch?v=mVqiCgaLSf4&app=desktop (last accessed 17.1.2020)
- ↑ Swan L, Dupont J. Multiple System Atrophy. Journal of Physical Therapy 1999;79:488-94.
- ↑ 10.0 10.1 10.2 MSA Coalition. MSA what you need to know 4.4.2019. Available from: https://www.multiplesystematrophy.org/wp-content/uploads/2019/09/What-You-Need-to-Know-9.4.19-DSC-AM-NV-Edits2.pdf (last accessed 17.1.2020)
- ↑ MAS coalition Depression and Cognitive Impairment in MSA Available:https://www.multiplesystematrophy.org/about-msa/depression-cognitive-impairment/ (accessed 14.3.2022)
- ↑ Wenning GK, Braune S. Multiple System Atrophy: Pathophysiology and Management. CNS Drugs 2001;15:839-48.
- ↑ 13.0 13.1 13.2 Wedge F. The Impact of Resistance Training on Balance and Functional Ability of a Patient with Multiple System Atrophy. Journal of Geriatric Physical Therapy 2008;31:79-83.
- ↑ 14.0 14.1 Hardy, Joanne. Multiple System Atrophy: Pathophysiology, Treatment and Nursing Care. Nursing Standard 2008;22:50-6.
- ↑ O'Sullivan SS, Massey LA, Williams DR, Silveira-moriyama L, Kempster PA, Holton JL, Revesz T, Lees, AJ. Clinical Outcomes of Progressive Supranuclear Palsy and Multiple System Atrophy. Brain. 2008;131:1362-72
- ↑ Hain TC. Multiple System Atrophy. 2010. http://www.dizziness-and-balance.com/disorders/central/movement/msa.html (accessed on 5 april 2010).