Myasthenia Gravis
Original Editor - Wendy Walker
Top Contributors - Laura Ritchie, Garima Gedamkar, Khloud Shreif, Wendy Walker, Nikhil Benhur Abburi, Rachael Lowe, Sheik Abdul Khadir, Evan Thomas, Lucinda hampton, Admin, Kim Jackson, Sai Kripa, Saeed Dokhnan, WikiSysop and Candace Goh
Introduction[edit | edit source]
Myasthenia gravis (MG) is an autoimmune disorder affecting the neuromuscular junction. It is a defect in transmission of nerve impulses to muscles at neuromuscular junction. It is a relatively rare, long term condition caused by circulating antibodies that block acetylcholine receptors at the postsynaptic neuromuscular junction, inhibiting the excitatory effects of the neurotransmitter acetylcholine and prevents muscle contraction. MG affects people at any age but it commonly to affects women under 40. It manifests as a generalized muscle weakness which can involve the respiratory muscles and can lead to a myasthenic crisis, which is a medical emergency[1]. [2]The below 5 minute video outlines the condition.
Clinically Relevant Anatomy[edit | edit source]
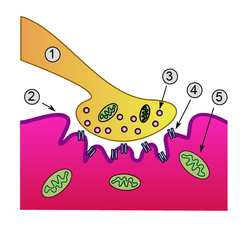
Detailed view of a neuromuscular junction:
- Presynaptic terminal
- Sarcolemma
- Synaptic vesicle
- Nicotinic acetylcholine receptor
- Mitochondrion
Epidemiology[edit | edit source]
The incidence rate of MG estimates to be about 5 to 30 cases per million person-years, and the prevalence rate to be between 10 to 20 cases per 100,000 population. There is equal geographic distribution in the incidence and prevalence of MG in both adults and children and juvenile MG that start before the age of 18 years is almost 10% of all cases of MG[4].
Its prevalence has been increasing over the past several decades secondary to better awareness, recognition, and increased survival, its not a linear increase, the age of onset is characterized by a bimodal distribution with an early incidence peak in the 2nd to 3rd decades affecting young women and a late peak in the 6th to 8th decades that is primarily seen in men[5].[6]
Mechanism of Injury / Pathological Process[edit | edit source]
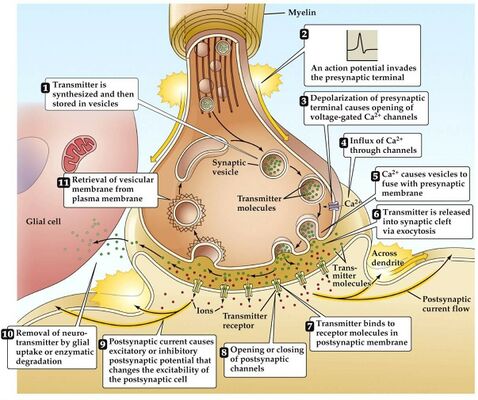
In Myasthenia gravis (MG) antibodies form against nicotinic acetylcholine (ACh) postsynaptic receptors at the neuromuscular junction (NMJ) of the skeletal muscles[7].The basic pathology is a reduction in the number of ACh receptors (AChRs) at the postsynaptic muscle membrane brought about by an acquired autoimmune reaction producing anti-AChR antibodies[8].
During normal conditions of depolarization Calcium channels on the presynaptic membrane open and triggering the release of ACh-R into the synaptic cleft when it reaches binds to postsynaptic membrane receptors where the end-plate potential (EPP) is generated, this potential is larger than the threshold needed to generate a postsynaptic action potential. In MG patients there is a decrease in the number or activity of ACh-R so decrease in the EPP, hence this potential may be sufficient at rest but with activities, there is not sufficient EPP to trigger an action potential.
Clinical Presentation[edit | edit source]
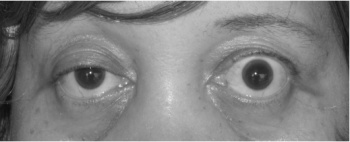
- The usual initial complaint is a specific muscle weakness rather than generalized weakness - frequently ocular (eye) symptoms.
- Extraocular muscle weakness or ptosis is present initially in 50% of patients and occurs during the course of illness in 90% of patients. Patients also frequently report diplopia (double vision).
- The disease remains exclusively ocular in 10 - 40% of patients.
- Rarely, patients have generalised weakness without ocular muscle weakness.
- Eye muscle weakness are often asymmetrical and variable.
- Bulbar muscle weakness is also common, fascial muscle weakness can result in, trouble smiling or trouble whistling.
- Weakness tends to spread from the ocular to facial to bulbar muscles and then to truncal and limb muscles.
- With the weakness of neck flexors more common and neck extensor weakness is rare to present.
- Limb weakness may be more severe proximally than distally.
- Isolated limb muscle weakness is the presenting symptom in fewer than 10% of patients.
- Weakness is typically least severe in the morning and worsens as the day progresses.
- Weakness is increased by exertion and alleviated by rest.
- Weakness progresses from mild to more severe over weeks or months, with exacerbations and remissions.
- About 87% of patients have generalised disease within 13 months after onset, and about 75% will have generalized weakness within the first 2 to 3 years.
- Less often, symptoms may remain limited to the extra-ocular and eyelid muscles for many years.
- Respiratory muscle weakness develop in up to 40% of patients and myasthenic crisis( respiratory failure) present in about 15%- 20% of patients with MG[4].
Classification[edit | edit source]
Subtypes of MG are broadly classified as follows:[9]
- Early-onset MG: age at onset <50 years. Thymic hyperplasia, usual females.
- Late-onset MG: age at onset >50 years. Thymic atrophy, mainly males.
- Thymoma-associated MG (10%–15%)
- MG with anti-MUSK antibodies.
- Ocular MG (oMG): symptoms only affecting extraocular muscles.
- MG with no detectable AChR and muscle-specific tyrosine kinase (MuSK) antibodies.
The most widely utilised classification of MG is the Myasthenia Gravis Foundation of America Clinical Classification[10]
- Class I: Any ocular muscle weakness, possible ptosis, no other evidence of muscle weakness elsewhere
- Class II: Mild weakness affecting other than ocular muscles; may also have ocular muscle weakness of any severity
- Class IIa: Predominantly affecting limb, axial muscles, or both; may also have lesser involvement of oropharyngeal muscles.
- Class IIb: Predominantly bulbar and/or respiratory muscles; may also have lesser or equal involvement of limb, axial muscles, or both.
- Class III: Moderate weakness affecting other than ocular muscles; may also have ocular muscle weakness of any severity
- Class IIIa: Predominantly affecting limb, axial muscles, or both; may also have lesser involvement of oropharyngeal muscles
- Class IIIb: Predominantly bulbar and/or respiratory muscles; may also have lesser or equal involvement of limb, axial muscles, or both
- Class IV: Severe weakness affecting other than ocular muscles; may also have ocular muscle weakness of any severity
- Class IVa: Predominantly affecting limb, axial muscles, or both; may also have lesser involvement of oropharyngeal muscles
- Class IVb: Predominantly bulbar and/or respiratory muscles; may also have lesser or equal involvement of limb, axial muscles, or both (Can also include feeding tube without intubation)
- Class V: Intubation needed to maintain airway, with or without mechanical ventilation
Diagnostic Procedures[edit | edit source]
The diagnosis of myasthenia gravis based on the associated and confirmed symptoms and signs the patient presented with plus a positive test for specific autoantibodies, which are important to detect MG and define MG subgroups.
1- The most commonly immunological tets:
Antibodies against acetylcholine receptors (Anti- AchR) is highly specific for MG, Anti- AchR not used to predict the severity of the disease, negative test results (Seronegativity) may occur if the test is done early or if it is immunosuppression condition. It has a sensitivity of approximately 85% for gMG and 50% for oMG.
Muscle-specific kinase antibodies antibodies.
Antibodies against striated muscle cytoplasmic proteins: found in thymoma patients with and without myasthenia gravis.
2- Tensilon (Edrophonium Chloride) Test: pharmacological test used for the diagnosis of certain neural diseases, mainly MG and distinguish Myasthenia crisis from the cholinergic crisis. It is An Ach-R inhibitor that prolongs the duration of Ach in NMJ.
Procedure: it is administered intravenously IV while the patient is observed for any objective improvement in muscle strength especially movement f extraocular muscles or improvement in ptosis, while asymmetrical improvement in eyelid muscles is considered a positive result for the test. During this test, the patients must be monitored for cardiac and blood pressure prior to injection because of the possible risk of arrhythmia and hypotension.
Side effects: increase in salivation, sweating, nausea, stomach cramping, and muscle fasciculation. While hypotension and bradycardia are uncommon side effects and relief with rest.
Sensitivity: Tensilon test has a sensitivity of 71.5%–95% for the diagnosis of MG.
3- Ice Pack Test: nonpharmacological test used when Edrophonium Chloride test is contraindicated, used for patients with ptosis. Apply an ice pack over the eye for 2–5 minutes and assessing for improvement in ptosis.
4- Electrophysiological Tests: using repetitive nerve stimulation study and single-fiber electromyography are the main electrophysiologic tests for diagnosis of MG. The nerve stimulated supramaximally at 2-3 Hz, and the decrement between the first and the fifth evoked muscle action potential is diagnostic for MG. It is abnormal in approximately 75% of patients with generalized myasthenia gravis and 50% of patients with Ocular myasthenia gravis (oMG).
Single-fiber electromyography (SFEMG) : it uses a needle electrode to identify AP from individual ms fibers and can record the action potential of muscle fibers innervated by the same motor axon. Although it is highly sensitive test abnormality didn’t mean it is necessary to be MG that may be another motor neuron disease, polymyositis, or peripheral neuropathy
Sensitivity: the sensitivity of this test is approximately 85% for gMG and 50% for oMG[9][11] .
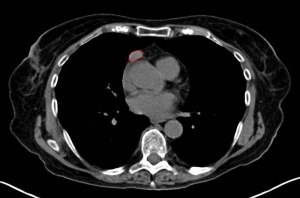
5- CT scan / MRI for thymoma investigation
Differential Diagnosis[edit | edit source]
- Amyotrophic Lateral Sclerosis
- Basilar Artery Thrombosis
- Brainstem Gliomas
- Cavernous Sinus Syndromes
- Dermatomyositis/Polymyositis
- Lambert-Eaton Myasthenic Syndrome
- Multiple Sclerosis
- Myocardial Infarction
- Pulmonary Embolism
- Sarcoidosis and Neuropathy
- Thyroid Disease
- Tolosa-Hunt Syndrome
Outcome Measures[edit | edit source]
Management / Interventions[edit | edit source]
Medical Management[edit | edit source]
There are four basic therapies used to treat MG:
- Symptomatic treatment with acetylcholinesterase inhibitors.
- Rapid short-term immunomodulating treatment with plasmapheresis and intravenous immunoglobulin.
- Chronic long-term immunomodulating treatment with glucocorticoids and other immunosuppressive drugs.
- Surgical treatment: thymectomy may be needed for cases with thymoma
Surgical Management (Thymectomy)[edit | edit source]
Thymus gland has a key role in inducing production of acetylcholine receptor antibody in patients with myasthenia gravis. Removal of the gland in cases with tumor (total thymectomy) showed better outcomes and patients will show improvement in symptoms, usage of immunosuppressive drugs, on the other side thymectomy is not recommended for patients with ocular myasthenia, since there is not sufficient evidence that surgery prevents generalization or results in remission.
Physiotherapy Management[edit | edit source]
Rehabilitation alone or in combination with other forms of treatment such as psychotherapy help to relieve or reduce symptoms for some people with MG. Despite the lack of high-quality evidence for effective rehabilitation modalities for people with MG, a spectrum of interventions are suggested and applied. These interventions help to reduce fatigue and to improve the quality of life, although an effective neuro rehabilitation program can control or prevent physical deformities and minimise secondary medical comorbidities.
Physical activity and physical training of low to medium intensity is recommended, while physical activity of high intensity should be avoided in patients with MG because it increases muscular weakness[13]. Individuals with MG should strive to strike a balance between physical activity and rest, finding the right level of each to optimise their health. It is not possible to cure the weakness by active physical training. However, most MG patients are more passive than they need to be.
One study showed a clear benefit from a strength training exercise program for a group of patients with mild to moderate MG[14], concluding "physical training can be carried out safely in mild MG and provides some improvement of muscle force".
Aerobic exercise, respiratory muscle training, strength training, and progressive resistance exercises has been shown to be an effective approach in enhancing functional outcomes (including mobility, muscle strength, and aerobic capacity), reducing fatigue, improving physical performance, and enhancing overall quality of life for individuals with MG[15]. Previous studies have reported that exercise programs for MG patients should be supervised to ensure safety[16]. However, a recent randomised controlled trial was done to explore effects of home-based, unsupervised exercise therapy in MG[17]. This the first study to report that a 3-month unsupervised home-based exercise therapy program was well-tolerated by adults with stabilised MG and did not caused any exacerbations. The therapy program did not show significant evidences on improvement in quality of life. Further research is needed to investigate different dosages of exercise and their effects in people living with MG.
- Balance strategy training maybe effective in improving balance, enhance the vestibular function through trgeting sensorimotor system to improve balance and decrease falling risk, moreover more research into this domain has to be done[18].
- Gradual resisted training exercises.
- Respiratory muscle training showed to be effective in management fatigable weakness and respiratory failure specially with moderate cases that hinder normal ADL. The benefits of respiratory training summarized in the following ( improvement in respiratory muscle strength and endurance, improvement in physical performance and reduce incidence of respiratory MG complications[19].
General advice for exercise programs for people with MG:
- Aim to strengthen large muscle groups, particularly proximal muscles of shoulders and hips
- Advise patient to do the exercises at their "best time of day" ie, when not feeling tired - for the majority of MG patients this will be morning
- If a patient is taking pyridostigmine, exercise at peak dose ie, 1.5 to 2 hours after taking a dose
- Moderate intensity of exercise only: the patient should not experience worsening of MG symptoms (eg. ptosis or diplopia) during exercise
- General aerobic exercise is also valuable, helping with respiratory function as well stamina.
Myasthenic Crisis[edit | edit source]
Myasthenic crisis is defined as respiratory muscle weakness that is severe enough to necessitate intubation or delay extubation. Prognosis in myasthenic crisis has dramatically improved over the last 4 decades from a mortality rate of 75% to the current 4.5%. It is prudent to closely observe MG patients with respiratory difficulty in a supervised setting. The forced vital capacity (FVC) and the negative inspiratory force (NIF) are the main respiratory parameters for monitoring, and both should be measured frequently during the hospital admission. Abnormalities of arterial blood gases are insensitive measures of respiratory muscle weakness as they often are late-occurring abnormalities only after the onset of life-threatening respiratory failure. Elective intubation should be considered if serial FVC measurements show values less than 20 mL/kg or if the NIF is less than 30 cm H2O.[20] Rapid induction therapies such as plasmapheresis or IVIg should be considered. In most cases, the initiation or maintenance of high-dose corticosteroids is also necessary.
Prognosis[edit | edit source]
With treatment, people with MG have a normal life expectancy. Some combination of medication, thymectomy, and other therapies enable most myasthenics to lead normal or near-normal lives. Sometimes people experience remission. However, for some people quality of life is affected significantly - either by the severity of the disease or severity of side effects from the medication. Generally, those who are quickly diagnosed and receive effective treatment have the best outcomes.
Medical treatment involves the use of anticholinesterase agents, immunosuppressive drugs, plasmapheresis, and gammaglobulin, with reported complete clinical remission rates (CCRRs) as low as 15%. Accordingly, thymectomy has become an increasingly accepted procedure for the treatment of MG, as it can achieve CCRRs as high as 80% in accordance with most of the reports published in the literature.
Resources[edit | edit source]
The Myasthenia Gravis Foundation of America (MGFA) has a comprehensive website
References[edit | edit source]
- ↑ Gilhus NE, Socrates T, Amelia E, Jacqueline P, Burns TM. Myasthenia gravis (Primer). Nature Reviews: Disease Primers. 2019;5(1).
- ↑ Suresh AB, Asuncion RM. Continuing Education Activity.2021 Available: https://www.ncbi.nlm.nih.gov/books/NBK559331/(accessed 4.5.2022)
- ↑ Osmosis. Myasthenia gravis - causes, symptoms, treatment, pathology. Available from: http://www.youtube.com/watch?v=bYGxGdu9MsQ[last accessed 10/9/2021]
- ↑ 4.0 4.1 Hehir MK, Silvestri NJ. Generalized myasthenia gravis: classification, clinical presentation, natural history, and epidemiology. Neurologic clinics. 2018 May 1;36(2):253-60.
- ↑ McGrogan A, Sneddon S, De Vries CS. The incidence of myasthenia gravis: a systematic literature review. Neuroepidemiology. 2010;34(3):171-83.
- ↑ Keesey JC. Clinical evaluation and management of myasthenia gravis. Muscle & Nerve: Official Journal of the American Association of Electrodiagnostic Medicine. 2004 Apr;29(4):484-505.
- ↑ Strauss AJ, Seegal BC, Hsu KC, Burkholder PM, Nastuk WL, Osserman KE. Immunofluorescence demonstration of a muscle binding, complement-fixing serum globulin fraction in myasthenia gravis. Proceedings of the Society for Experimental Biology and Medicine. 1960 Oct;105(1):184-91.
- ↑ Patrick J, Lindstrom J. Autoimmune response to acetylcholine receptor. Science. 1973 May 25;180(4088):871-2.
- ↑ 9.0 9.1 Annapurni Jayam Trouth et. al. Autoimmune DiseasesfckLRVolume 2012 (2012), Article ID 874680, 10 pages
- ↑ Jaretzki A 3rd, Barohn RJ, Ernstoff RM, et al. Myasthenia gravis: recommendations for clinical research standards. Task Force of the Medical Scientific Advisory Board of the Myasthenia Gravis Foundation of America. Neurology. Jul 12 2000;55(1):16-23
- ↑ Sieb JP. Myasthenia gravis: an update for the clinician. Clinical & Experimental Immunology. 2014 Mar;175(3):408-18.
- ↑ Connie Koklanis. Myasthenia Gravis Clinical Investigations. Available from: http://www.youtube.com/watch?v=2CxQeal8xiQ[last accessed 10/11/2021]
- ↑ Skeie GO, Apostolski S, Evoli A et al. Guidelines for treatment of autoimmune neuromuscular transmission disorders. Eur. J. Neurol.17,893–902 (2010)
- ↑ Lohi EL1, Lindberg C, Andersen O. Physical training effects in myasthenia gravis. Arch Phys Med Rehabil. 1993 Nov;74(11):1178-80
- ↑ Corrado B, Giardulli B, Costa M. Evidence-based practice in rehabilitation of myasthenia gravis. A systematic review of the literature. Journal of Functional Morphology and Kinesiology. 2020 Sep 27;5(4):71.
- ↑ Westerberg, Molin CJ, Lindblad I, Emtner M, Punga AR. Physical exercise in myasthenia gravis is safe and improves neuromuscular parameters and physical performance-based measures: A pilot study. Muscle Nerve 56: 207-214, 2017
- ↑ Birnbaum S, Porcher R, Portero P, Clair B, Demeret S, Eymard B, Gargiulo M, Louët E, Berrih-Aknin S, Le Panse R, Aegerter P. Home-based exercise in autoimmune myasthenia gravis: a randomized controlled trial. Neuromuscular Disorders. 2021 Aug 1;31(8):726-35.
- ↑ S H Wong, J Nitz, K Williams, S Brauer Effects of balance strategy training in myasthenia gravis: a case study series May 2015 Volume 101, Supplement 1, Pages e1657–e1658
- ↑ Corrado B, Giardulli B, Costa M. Evidence-Based Practice in Rehabilitation of Myasthenia Gravis. A Systematic Review of the Literature. Journal of Functional Morphology and Kinesiology. 2020 Dec;5(4):71.
- ↑ Jani-Acsadi A, Lisak RP. Myasthenic crisis: guidelines for prevention and treatment. Journal of the neurological sciences. 2007 Oct 15;261(1-2):127-33.