Neonatal Respiratory Distress Syndrome
Original Editor - Lucinda hampton
Top Contributors - Lucinda hampton
Introduction[edit | edit source]
Neonatal respiratory distress syndrome, or RDS, is a common cause of respiratory distress in a newborn, presenting within hours after birth, most often immediately after delivery. RDS primarily affects preterm neonates, and infrequently, term infants. The incidence of RDS is inversely proportional to the gestational age of the infant, with more severe disease in the smaller and more premature neonates. While treatment modalities, including antenatal corticosteroids, surfactants, and advanced respiratory care of the neonate, have improved the outcomes for patients affected by RDS, it continues to be a leading cause of morbidity and mortality in the preterm infant[1].
- Early respiratory distress syndrome can be caused by COVID-19. A 2020 case report from the researchers at the University of São Paulo, published in the journal CLINICS, shows how newborns can also be infected with COVID-19 and develop serious symptoms eg RDS[2].Vertical transmission seems possible but has not been documented[2]
Epidemiology[edit | edit source]
The incidence is estimated at 6 per 1000 births. Uncommon after 36 weeks' gestation due to development of pneumocyte surfactant production around 35 weeks[3].
In one study of babies born between 2003 and 2007 at various National Institute of Child Health and Human Development (NICHD) Neonatal Research Network centers, 98% of babies born at 24 weeks had RDS, while at 34 weeks, the incidence was 5%, and at 37 weeks was less than 1%[1]
Clinical presentation[edit | edit source]
Respiratory distress presents in the first few hours of life in a premature baby. Signs include tachypnoea, expiratory grunting, and nasal flaring. The infant may or may not be cyanosed. Substernal and intercostal retractions may be evident.
- Risk factors include maternal diabetes, greater prematurity, perinatal asphyxia and multiple gestations.
- Associated abnormalities are those that can occur in prematurity: germinal matrix haemorrhage, necrotising enterocolitis, patent ductus arteriosus, delayed developmental milestones, hypothermia and hypoglycaemia[3].
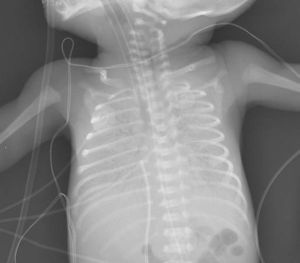
Image: Preterm neonate with complete opacification of both hemithoraces. Features of RDS.
Pathology[edit | edit source]
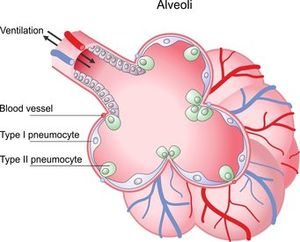
Immature type II pneumocytes (described here) cannot produce surfactant. The lack of surfactant increases the surface tension in alveoli causing collapse. Patients have a decreased lecithin to sphingomyelin ratio (a method for clinicians to assess fetal lung maturation, a sample of amniotic fluid is collected via amniocentesis to determine the risk of the neonate developing RDS[4]). Damaged cells, necrotic cells, and mucus line the alveoli.
- Although most cases are related to prematurity alone, rarely patients may have genetic disorders of surfactant production and can present in a similar clinical and radiological manner.
- As the alveoli are collapsed (microscopically), the lungs are collapsed macroscopically. It is a diffuse type of adhesive atelectasis
Evaluation[edit | edit source]
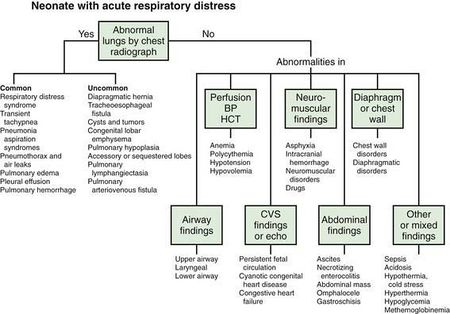
Since the definition of neonatal respiratory distress syndrome is imprecise, prompt diagnosis and treatment require an overall assessment of prenatal and delivery history to identify perinatal risk factors, clinical presentation, radiographic findings, and evidence of hypoxemia on blood gas analysis. The clinical presentation consists of non-specific respiratory symptoms, including tachypnea, nasal flaring, grunting, retractions, and cyanosis, with decreased air entry on auscultation.
- Chest Radiography: pathognomonic of RDS include homogenous lung disease with diffuse atelectasis.
- Arterial Blood Gas Analysis: may show hypoxemia that responds to increased oxygen supplementation and hypercapnia. Serial blood gases may show evidence of worsening respiratory and metabolic acidosis, including lactic acidemia in infants with worsening RDS.
- Other Investigations: eg. An echocardiogram may show the presence of a patent ductus arteriosus that might complicate the clinical course of RDS.[1].
Treatment / Management[edit | edit source]
The goals of optimal management of neonatal respiratory distress syndrome include decreasing incidence and severity using antenatal corticosteroids, followed by optimal management using respiratory support, surfactant therapy, and overall care of the premature infant.
- Antenatal corticosteroids
- Monitoring oxygenation and ventilation
- Assisted ventilation of the neonate (see below)
- Exogenous surfactant therapy: Exogenous animal-derived or synthetic pulmonary surfactant therapy has already shown immense success in the treatment of neonatal respiratory distress syndrome and has the potential to contribute efficiently toward repair of damaged alveoli and preventing severe acute respiratory syndrome coronavirus 2-associated respiratory failure.[5]
- Supportive care, including thermoregulation, nutritional support, fluid and electrolyte management, antibiotic therapy, etc[1]
Ventilation[edit | edit source]
Once resuscitated and given surfactant (if appropriate), infants should be assessed. Depending on maturity and severity of the RDS, their care needs could range from basic nursing care in an incubator with or without humidified oxygen, through oxygen therapy via a nasal cannula, to intensive care with mechanical ventilation
Assisted Ventilation of the Neonate
The goals of assisted ventilation are to reduce atelectasis by providing a constant distending positive airway pressure. The current preferred strategy is the early initiation of continuous positive airway pressure (CPAP) with selective surfactant administration. In most institutions, non-invasive modalities are preferred over invasive ventilation as they decrease the risk of mortality, and bronchopulmonary dysplasia (BPD) compared to invasive ventilation with or without surfactant
- Continuous Positive Airway Pressure (CPAP): Nasal CPAP is an initial intervention in preterm infants with RDS or risk of RDS without respiratory failure. Multiple modalities are available for CPAP delivery, including ventilator derived CPAP as well as a less expensive bubble CPAP device. Infants who received CPAP fared as well as infants who received prophylactic surfactant therapy along with mechanical ventilation in the SUPPORT trial (Surfactant Positive Airway Pressure and Pulse Oximetry Randomized Trial), and those who received early CPAP had a reduced need for surfactant therapy. Also, the incidence of BPD decreased with the use of CPAP. The goals of treatment include keeping SpO2 between 90-95%, and PaCO2 between 45-65 mmHg.
- Non-invasive Respiratory Support: Nasal Intermittent Positive Pressure Ventilation (NIPPV) appears superior to CPAP alone for decreasing extubation failure, the need for intubation in preterm infants, but the same in cost and safety. The primary difference in NIPPV and CPAP is that NIPPV requires a ventilator to provide positive pressure ventilation, while CPAP may use a less expensive device such as bubble CPAP to deliver the appropriate pressures.
- High Flow Nasal Canula: Heated humidified high-flow nasal cannulas (HFNC) are also used in some centers as an alternative to CPAP to provide positive distending pressure ventilation to neonates with RDS. As seen in a clinical trial by Roberts et al., HFNC was found to be inferior to CPAP.
- Mechanical Ventilation: Patients who do not respond to CPAP, develop respiratory acidosis (PH < 7.2 and PaCo2 > 60-65 mm of Hg), hypoxemia (PaO2 < 50 mm of Hg or Fio2 > 0.40 on CPAP), or severe apnea are managed with endotracheal intubation and mechanical ventilation. The goals of mechanical ventilation include providing adequate respiratory support while balancing the risks of barotrauma, volutrauma, and oxygen toxicity[1][6].
Prognosis[edit | edit source]
Prognosis of infants managed with antenatal steroids, respiratory support, and exogenous surfactant therapy is excellent.
- Mortality is less than 10%, with some studies showing survival rates of up to 98% with advanced care. Increased survival in developed countries is in stark comparison to babies who received no intervention in low-income countries, where the mortality rate for premature infants with RDS is significantly higher, at times close to 100%.
- With adequate ventilatory support alone, surfactant production eventually begins, and once surfactant production begins along with the onset of diuresis, RDS improves within 4 or 5 days.
- Untreated disease leading to severe hypoxemia in the first days of life can result in multiple organ failure and death[1].
Differential diagnosis[edit | edit source]
Consider:
- congenital heart disease
- group B Streptococcal pneumonia
- pulmonary haemorrhage
- pulmonary oedema / pulmonary venous congestion
- neonatal pneumonia
- transient tachypnoea of the newborn: lung volumes are normal to slightly hyperinflated in TTN and decreased in RDS[3]
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Yadav S, Lee B, Kamity R. Neonatal Respiratory Distress Syndrome. StatPearls [Internet]. 2020 Jul 19.Available : https://www.ncbi.nlm.nih.gov/books/NBK560779/ (accessed 18.4.2021)
- ↑ 2.0 2.1 Carvalho WB, Gibelli MA, Krebs VL, Calil VM, Nicolau CM, Johnston C. Neonatal SARS-CoV-2 infection. Clinics. 2020;75.Available: https://www.clinicsjournal.com/article/neonatal-sars-cov-2-infection/ (accessed 18.4.2021)
- ↑ 3.0 3.1 3.2 Radiopedia IRDS Available from: https://radiopaedia.org/articles/respiratory-distress-syndrome(accessed 18.4.2021)
- ↑ Ogbejesi C, Tadi P. Lecithin Sphingomyelin Ratio. StatPearls [Internet]. 2020 Jul 8.Available : https://pubmed.ncbi.nlm.nih.gov/32809485/ (accessed 18.4.2021)
- ↑ Ghati, A., Dam, P., Tasdemir, D., Kati, A., Sellami, H., Sezgin, G.C., Ildiz, N., Franco, O.L., Mandal, A.K. and Ocsoy, I., 2021. Exogenous pulmonary surfactant: A review focused on adjunctive therapy for severe acute respiratory syndrome coronavirus 2 including SP-A and SP-D as added clinical marker. Curr Opin Colloid Interface Sci, 51, p.101413.Available :https://pubmed.ncbi.nlm.nih.gov/33390762/ (accessed 18.4.2021)
- ↑ Boyd S. Causes and treatment of neonatal respiratory distress syndrome. Nursing times. 2004 Jul 1;100(30):40-4.Available from:https://www.nursingtimes.net/clinical-archive/respiratory-clinical-archive/causes-and-treatment-of-neonatal-respiratory-distress-syndrome-27-07-2004/ (accessed 18.4.2021)