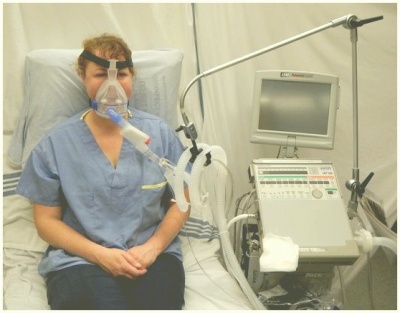
Non Invasive Ventilation
Original Editor - The Open Physio project. Top Contributors - Kim Jackson, George Prudden, Admin, Rachael Lowe, Tomer Yona, WikiSysop, Vidya Acharya and Lucinda hampton
Introduction[edit | edit source]
Non-invasive ventilation (NIV) is the delivery of oxygen (ventilation support) via a face mask and therefore eliminating the need of an endotracheal airway.[1] NIV achieves comparative physiological benefits to conventional mechanical ventilation by reducing the work of breathing and improving gas exchange.[2] Research suggests that noninvasive ventilation after early extubation looks helpful in reducing the total days spent on invasive mechanical ventilation.[3]
The intervention is recognised as an effective treatment for respiratory failure in chronic obstructive pulmonary disease, cardiogenic pulmonary oedema and other respiratory conditions without complications such as respiratory muscle weakness, upper airway trauma, ventilator-associated pneumonia, and sinusitis.[1][4]
NIV works by creating a positive airway pressure - the pressure outside the lungs being greater than the pressure inside of the lungs. This causes air to be forced into the lungs (down the pressure gradient), lessening the respiratory effort and reducing the work of breathing[5]. It also helps to keep the chest and lungs expanded by increasing the functional residual capacity (the amount of air remaining in the lungs after expiration) after a normal (tidal) expiration; this is the air available in the alveoli available for gaseous exchange[6]. There are two types of NIV non-invasive positive-pressure (NIPPV) and Negative-Pressure Ventilation (NPV).
For anatomy of the lungs see here
Non-Invasive Positive-Pressure[edit | edit source]
NIPPV describes the delivery of oxygen at either constant or variable pressures via a face mask, such as Bi-level Positive Airway Pressure (BiPAP) and Constant Positive Airway Pressure (CPAP)
CPAP[edit | edit source]
CPAP is the most basic level of support and provides constant fixed positive pressure throughout inspiration and expiration, causing the airways to remain open and reduce the work of breathing[8]. This results in a higher degree of inspired oxygen than other oxygen masks. When indicated for home use it is usually via a low flow generator and is commonly used for patients requiring nocturnal CPAP for sleep apnoea[9]. High flow systems used in a hospital environment are designed to ensure that airflow rates delivered are greater than those generated by the distressed patient[9]. As well as having an effect on respiratory function it can also assist cardiac function where patients have a low cardiac output with pre-existing low blood pressure[9]. It is also commonly used for severe obstructive sleep apnoea and also for type 1 respiratory failure, for example, acute pulmonary oedema (by recruiting collapsed alveoli).
Indications[edit | edit source]
- When a patient remains hypoxic despite medical intervention
- Atelectasis - Complete or partial collapse of a lung or lobe[10]
- Rib fractures - to splint the rib cage open; to stabilise the fracture and prevent damage to the lung[10]
- Type I respiratory failure
- Congestive Heart Failure
- Cardiogenic pulmonary oedema
- Obstructive sleep apnoea
- Pneumonia: as an interim measure before invasive ventilation or as a ceiling of treatment
- Nasal CPAP is more commonly used with infants.[11]
BiPAP[edit | edit source]
NIV is often described as BiPAP, however, BiPAP is actually the trade name. As the name suggests provides differing airway pressure depending on inspiration and expiration. The inspiratory positive airways pressure (iPAP) is higher than the expiratory positive airways pressure (ePAP)[12]. Therefore, ventilation is provided mainly by iPAP, whereas ePAP recruits underventilated or collapsed alveoli for gas exchange and allows for the removal of the exhaled gas. In the acute setting, NIV is used in type 2 respiratory failure (for example in a COPD exacerbation), with respiratory acidosis (pH < 7.35)[13].
Indications[edit | edit source]
- Type II respiratory failure
- Acidotic exacerbation of chronic obstructive pulmonary disease (COPD)[14]
- Increased work of breath causing ventilatory failure, for example, hypercapnia (increased CO2 in arterial blood gas), fatigue or neuromuscular disorder
- Weaning from tracheal intubation
Negative-Pressure Ventilation (NPV)[edit | edit source]
Negative-pressure ventilators provide ventilatory support using a device that encases the thoracic cage, such as the iron lung. Although not seen as much in today's society they were popular in the first half of the twentieth century during the polio epidemic. They work by lowering the pressure surrounding the thorax, creating subatmospheric pressure which passively expands the chest wall to inflate the lungs. Exhalation occurs with passive recoil of the chest wall. Their use is still indicated in chronic respiratory failure. The three types used each with their own advantages and disadvantages[15]:
- The earliest version is the tank ventilator, more commonly known as the iron lung. It is a large cylindrical device that encases the patient's body with only the head visible, a neck collar provides an airtight seal
- The poncho-wrap is an airtight bodysuit using a rigid metal framework covered with an airtight nylon parker that surrounds the trunk
- The cuirass is made up of a rigid fibreglass shell which fits over the chest wall and upper abdomen
Contraindications of NIV[edit | edit source]
- Coma
- Undrained pneumothorax
- Frank haemoptysis
- Vomiting blood (haematemesis)
- Facial fractures
- Cardiovascular system instability
- Cardiac Arrest
- Respiratory Failure
- Raised ICP
- Recent upper GI surgery
- Active Tuberculosis
- Lung abscess
- No additional contraindications in the paediatric population[11]
Precautions[edit | edit source]
Patients Unlikely to Do Well On NIV[edit | edit source]
- Agitation, encephalopathic, uncooperative
- Severe illness including extreme acidosis (pH <7.2)
- Presence of excessive secretions or pneumonia
- Multiple organ failure
- Haemodynamic instability
- Inability to maintain a lip seal
- Inability to protect the airway
- Overt respiratory failure requiring immediate intubation[11]
Setting Up the Equipment[edit | edit source]
- Do not set up NIV unless you are familiar with the equipment, circuits, masks, etc.and are confident as to how safely to establish the patient on NIV and appropriately respond to blood gas results.
- The decision to use NIV and the settings must always be made with the medial and nursing team looking after the patient.
- Introduce the treatment to the patient slowly.
- Patients need to keep their mouth closed if using a nasal mask.
- Some patients are less suited to NIV; however, each situation should be individually assessed.
- NIV should generally be used in ICU/HDU environments - make sure you are aware of your local policy.[11]
Parts of the Machine[edit | edit source]
- Bi-level positive airway pressure (BiPAP) generator
- Anti-bacterial filter
- Smoothbore tubing
- Exhalation port
- Face mask, spacer and headgear
- Oxygen tubing
- Heated humidifier and tubing (if required)
- Oximeter with an integral recorder
Instructions on Using the Ventilator[edit | edit source]
- Introduce the patient slowly to the equipment and all its parts.
- Ensure the mask fits comfortably and that the patient can experience the mask on their face without the ventilator.
- Allow the patient the opportunity to feel the operation of the machine through the mask on their hands or cheek before applying it over their nose or mouth.
- Allow the patient the opportunity to practice breathing with the ventilator, either by holding the mask in place or allowing them to hold it in place.
- Adjust the settings initially for comfort and establish whether the patient can relax comfortably in a sleeping position.
- Provide opportunities for the patient to feedback any discomfort.
- Assess and adjust the performance of the ventilator during an afternoon nap to optimise gas exchange and comfort.
- Progress to an overnight study, continuing to monitor and optimise gas exchange and sleep quality.[11]
Complications[edit | edit source]
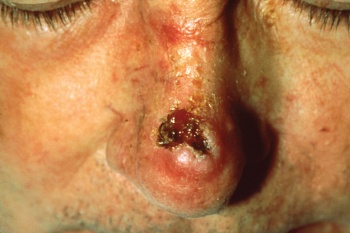
Facial Pressure Ulcers[edit | edit source]
Pressure ulcers associated with the use of NIV is a growing clinical problem due to the increased popularity of the intervention. Prevalence of grade I pressure ulcers have been estimated at 5-50% after a couple of hours and 100% after 48 hours[17]. The development of pressure ulcers is associated with poor clinical outcomes, increased complications, and length of hospital stay that compound with the consequences of acute illness. Medical devices such as NIV masks have unique risk factors including - the existence of a microclimate individual to the device, the method in which the device is secured, that devices may obscure the skin, and that the areas at risk are not routinely checked[18]. Clinicians’ primary focus has been to attain a mask seal, as air leaks are associated with reduced tolerance to the intervention[19]. The alternating airflow from bi-level positive pressure means that a seal is important to avoid ventilator asynchrony. Therefore strap tension is increased, with the risk of pressure damage a secondary consideration[20]. It is important to consider that the patient may not be able to respond to an uncomfortable mask fit or excessive load delivered to vulnerable areas of skin due to sedation, medication, or neurological disease or injury. Furthermore, the patient may be too weak to reposition the device. Oronasal masks have traditionally been preferred for their comfort and ease of use however other interfaces have been recommended as superior[21]. Prophylactic interventions should also be considered[22].
Eye Irritation[edit | edit source]
It is important to ensure the mask is fitted correctly if it is not it can cause oxygen to leak upwards to the eyes, causing eye irritation and conjunctivitis.[23]
Retention of Secretions[edit | edit source]
The use of a full face mask may interfere with the ability to cough and the effective clearance of secretions. As well as this, the positive pressure created may compromise the patient's ability to generate sufficient expiratory flow rates affecting the mobilisation of secretions and also a resistance to cough leading to the retention of secretions.[9]
Resources[edit | edit source]
References[edit | edit source]
- ↑ 1.0 1.1 Nava S, Hill N. Non-invasive ventilation in acute respiratory failure. Lancet 2009; 374(9685): 250-9.
- ↑ Vitacca M, Ambrosino N, Clini E, et al. Physiological Response to Pressure Support Ventilation Delivered before and after Extubation in Patients Not Capable of Totally Spontaneous Autonomous Breathing. American Journal of Respiratory and Critical Care Medicine 2001; 164: 638-41.
- ↑ Vaschetto R, Pecere A, Perkins GD, Mistry D, Cammarota G, Longhini F, Ferrer M, Pletsch-Assunção R, Carron M, Moretto F, Qiu H. Effects of early extubation followed by noninvasive ventilation versus standard extubation on the duration of invasive mechanical ventilation in hypoxemic non-hypercapnic patients: a systematic review and individual patient data meta-analysis of randomized controlled trials. Critical Care. 2021 Dec;25(1):1-1.
- ↑ Pingleton SK. Complications of acute respiratory failure. Am Rev Respir Dis 1988; 137(6): 1463-93.
- ↑ Guideline BT. Non-invasive ventilation in acute respiratory failure. Thorax. 2002 Mar;57(3):192-211.
- ↑ Lumb A. Nunn’s Applied Respiratory Physiology. 6th edn. Philidelphia PA: Butterworth Heinemann, 2005
- ↑ CPAP and Non-Invasive Ventilation in 5 minutes. Available at https://youtu.be/OHQK5PUTQ_0. Accessed 19 March 2020
- ↑ Pinto VL, Sharma S. Continuous Positive Airway Pressure (CPAP). InStatPearls [Internet] 2019 Jan 20. StatPearls Publishing. Accessed 19 March 2020
- ↑ 9.0 9.1 9.2 9.3 Nehyba, K. (2006). Continuous positive airway pressure ventilation part one: Physiology and patient care. British Journal of Cardiac Nursing, 1(12), 575–579. doi:10.12968/bjca.2006.1.12.22455
- ↑ 10.0 10.1 Nehyba, K. (2007). Continuous positive airway pressure ventilation. Part two: Indications and contraindications. British Journal of Cardiac Nursing, 2(1), 18–24. doi:10.12968/bjca.2007.2.1.22638
- ↑ 11.0 11.1 11.2 11.3 11.4 11.5 Harden, B. (2004). Emergency physiotherapy: An on-call survival guide. Edinburgh: Churchill Livingstone, pp.253-254.
- ↑ Hörmann C, Baum M, Putensen CH, Mutz NJ, Benzer H. Biphasic positive airway pressure (BIPAP) - A new mode of ventilatory support. European journal of anaesthesiology. 1994 Jan;11(1):37-42.
- ↑ Christie G, Currie GP, Plant P. Ventilatory support. Bmj. 2006 Jul 13;333(7559):138-40.
- ↑ Pertab D. Principles of non-invasive ventilation: a critical review of practice issues. British Journal of Nursing. 2009 Sep 10;18(16):1004-8.
- ↑ Shenoy KV, Kim V, Criner GJ. Noninvasive Ventilation. In Critical Care Study Guide 2010 (pp. 879-901). Springer, New York, NY.
- ↑ How to Set Up a BiPAP Machine. Available at https://www.youtube.com/watch?v=hXtx0nEoL9E. Accessed on 19 March 2020
- ↑ Carron M, Freo U, BaHammam AS, et al. Complications of non-invasive ventilation techniques: a comprehensive qualitative review of randomized trials. Br J Anaesth 2013; 110(6): 896-914.
- ↑ Black JM, Cuddigan JE, Walko MA, Didier LA, Lander MJ, Kelpe MR. Medical device related pressure ulcers in hospitalized patients. Int Wound J 2010; 7(5): 358-65.
- ↑ Dellweg D, Hochrainer D, Klauke M, Kerl J, Eiger G, Kohler D. Determinants of skin contact pressure formation during non-invasive ventilation. J Biomech 2010; 43(4): 652-7.
- ↑ Yamaguti WP, Moderno EV, Yamashita SY, et al. Treatment-related risk factors for development of skin breakdown in subjects with acute respiratory failure undergoing noninvasive ventilation or CPAP. Respir Care 2014; 59(10): 1530-6.
- ↑ Vaschetto R, De Jong A, Conseil M, et al. Comparative evaluation of three interfaces for non-invasive ventilation: a randomized cross-over design physiologic study on healthy volunteers. Crit Care 2014; 18(1): R2.
- ↑ Weng MH. The effect of protective treatment in reducing pressure ulcers for non-invasive ventilation patients. Intensive Crit Care Nurs 2008; 24(5): 295-9.
- ↑ Moore T, Woodrow P. High dependency nursing care: observation, intervention and support for level 2 patients. Routledge; 2009 Jun 4.