Complement System
Original Editor - Lucinda hampton
Top Contributors - Lucinda hampton, Kim Jackson and Nupur Smit Shah
Introduction[edit | edit source]
The complement system helps or “complements” the ability of antibodies and phagocytic cells to clear pathogens from an organism. It is part of the innate immune system.
The complement system consists of a number of small proteins found in the blood, made by the liver. Normally they circulate as inactive precursors. When stimulated by a trigger, proteases split these small proteins to release active cytokines. This starts a series (a cascade) of further cleavages which release more cytokines. This amplifies the response. So, if the original stimulus was an invading bacterium, the cytokines disrupt the phospholipid bilayer cell membrane of the target, which kills it [1].[2][3]
Complements are soluble proteins and glycoproteins, more than 20 types of complements are present in serum, found circulating normally in human body in inactive forms.
Complements are activated only during inflammatory reactions. During the inflammation, more amount of complements reaches to the interstitial area of the infected tissue through dilated blood vessels, which are then activated by proteolytic cleavage[1].
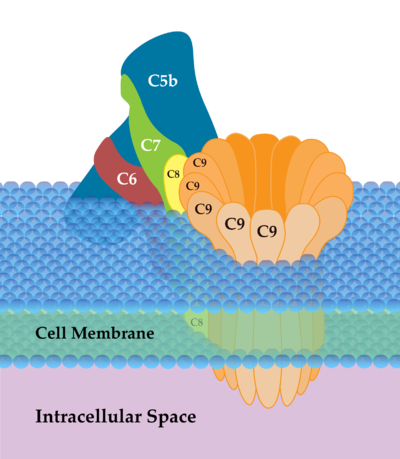
Image: Membrane Attack Complex (MAC) is the terminal complex of the complement system of the innate immune system. It forms pore-like holes in cell membranes, causing swelling that ultimately leads to cell death
Main functions of the Complement System[edit | edit source]
- Opsonization: Increases phagocytosis by opsonins (C4b and C3b) binding to foreign organisms Image 2: Phagocytosis
- Chemotaxis: Attracts macrophages and neutrophils via inflammation by inflammatory mediators; C5a, and to a lesser extent C3a and C4a
- Cell lysis: Ruptures membranes due to formation of a membrane attack complex (MAC)
- Agglutination: Causes clustering and binding of pathogens[4]
Pathways[edit | edit source]
Complement proteins in the circulation are not activated until triggered by an encounter with a bacterial cell, a virus, an immune complex, damaged tissue or other substance not usually present in the body.
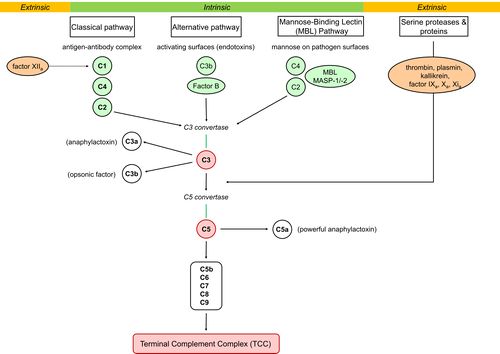
Image: A schematic representation of activation of 3 intrinsic pathways (classical, alternative, and mannose-binding lectin) to activate the complement cascade ie key-players C3 and C5 triggering the terminal pathway to constitute the Terminal Complement Complex (also known as MAC).
- Complement activation is a cascading event like the falling of a row of dominoes. It must follow a specific order if the end result is to be achieved.
- The circulating proteins have been grouped into three activation pathways, based on the types of substances and proteins that initiate the activation ie Classical; Lectin; Alternative[5]
- If you visualize a trident, the three tines represent the different initiation routes, while the handle represents the lytic mechanism by which this cascade ultimately destroys the threat, no matter which activation pathway started the response (all pathways converge to activate the pivotal protein of the complement system, called C3).[6]
Although they differ in mechanisms, the commonly needed step of all pathways is the conversion of C3 to C3a and C3b; the latter is necessary for the formation of the membrane attack complex (MAC).
- A goal of the complement system is the formation of MAC, which compromises the pathogen’s cell wall, causing swelling that ultimately leads to cell death[7].
Image 4: proteolytic cleavage
Complement Regulation[edit | edit source]
The complement system has the potential to be extremely damaging to host tissues; hence regulatory mechanisms are required to restrict the complement pathway. Various plasma and cell membrane proteins regulate complement activation by inhibiting different steps in the cascade[1].
Image 5: A complement protein attacking an invader.
Disease[edit | edit source]
The complement system is diffusely active within the body, and deficiencies or dysregulation results in immune system deficiencies, autoimmune disorders, or bleeding disorders[6].
- The complement system plays a critical role in inflammation and defence against some bacterial infections.
- Complement may also be activated during reactions against incompatible blood transfusions, and during the damaging immune responses that accompany autoimmune disease.
Complement deficiencies and defects eg:
- Deficiency in C1, C2, C3, MBL, MBL-associated serine protease 2 (MASP-2), factor H, factor I, or complement receptor 2 (CR2): Susceptibility to recurrent bacterial infections
- Deficiency of C5, C9, factor B, factor D, or properdin: Susceptibility to neisserial infections
- Defects in C1, C4, and C5: Systemic lupus erythematosus
- Defects in CR2: Common variable immunodeficiency
- Defects of CR3: Leukocyte adhesion deficiency type 1
- Mutations in the genes for factor B, factor H, factor I, membrane cofactor protein (CD46), or C3: Development of the atypical variant of hemolytic uremic syndrome[5]
References[edit | edit source]
- ↑ 1.0 1.1 1.2 Learn microbiology Complement Pathways: Types, Functions and Regulation Available from: https://microbeonline.com/complement-system-pathways-functions-regulation/(accessed 15.1.2021)
- ↑ Wisegeek Complimentary System Available from: https://www.wise-geek.com/what-is-the-complement-system.htm(accessed 15.1.2021)
- ↑ Kidzsearch Complement System Available from: https://wiki.kidzsearch.com/wiki/Complement_system (accessed 16.1.2021)
- ↑ Biorad Complement System mini review Available from: https://www.bio-rad-antibodies.com/complement-system-minireview.html?JSESSIONID_STERLING=F5D16F81B71103374705771E03996385.ecommerce1&JSESSIONID=D345BCCB6B7645656A89D67C223F28E9.lsds-e1a&evCntryLang=AU-en&cntry=AU&thirdPartyCookieEnabled=true (accessed 16.1.2021)
- ↑ 5.0 5.1 MDS manual Complement Available from:https://www.msdmanuals.com/en-kr/professional/immunology-allergic-disorders/biology-of-the-immune-system/complement-system (accessed 15.1.2021)
- ↑ 6.0 6.1 Britannica Complement Available from: https://www.britannica.com/science/complement-immune-system-component(accessed 15.1.2021)
- ↑ Haddad A, Wilson AM. Biochemistry, complement. StatPearls [Internet]. 2020 Sep 8.Available from: https://www.ncbi.nlm.nih.gov/books/NBK544229/#article-19783.s2(accessed 15.1.2021)