The Management of Parkinson’s Disease Symptoms with Tai Chi Exercise Program: A Case Study
The following case is entirely fictional, and was created for education purposes only.
Abstract[edit | edit source]
The objective of this case study was to explore the effectiveness of a Tai Chi exercise program in a patient who presents with symptoms consistent with the early to middle stages of Parkinson’s Disease . Recent evidence has shown optimistic findings in the use of Tai Chi to improve balance and gait impairments in patients with motor disorders such as PD.
The patient, Mrs. A is a 67 year-old retiree presenting with rigidity, bradykinesia and impaired gait and balance secondary to PD. She self-referred to physiotherapy to seek help with her recently worsening symptoms. Upon completion of a 24-week Tai Chi exercise program, Mrs. A showed improvements in postural stability and balance, gait pattern, bradykinesia symptoms, and ability to perform activities of daily living (ADLs) independently following completion of the program. She was also agreeable to continue doing Tai Chi following her discharge from physiotherapy after experiencing the changes in her level of motor function and confidence to remain independent. Outcome measures indicated that the Tai Chi exercise program provided benefits in some aspects of the patient’s emotional and physical well-being, however no effects were observed in overall posture, upper extremity tremors or rigidity. Additional research is needed to determine the most effective physiotherapy approaches for the management of PD, as well as longitudinal studies to observe the long-term effects of physiotherapy interventions.
Introduction[edit | edit source]
PD is a progressive neurological disorder that can cause deficits in all areas of life, including physical, cognitive, emotional, sleep-related, and systemic symptoms.[1] Furthermore, long-term use of medication to combat these symptoms can result in additional complications such as psychiatric side effects, dyskinesia, and motor fluctuations.[2] Parkinson's is the most common disorder of its type after Alzheimer’s disease, and prognosis and disease progression may vary greatly between individuals.[2] The first symptoms to appear are often motor, such as rigidity, bradykinesia, and tremors, which commonly lead to difficulties with balance and gait, thus increasing the risk for falls.[1] A report by Statistics Canada in 2014 suggests that diagnosis is not made until an average of almost two years after the appearance of initial symptoms,[2] therefore physical function is likely to have already deteriorated to some extent by the time physiotherapy treatment is initiated.
Although experts agree that exercise is an important part of Parkinson's management to preserve function and minimize the consequences of decreased mobilization, there is currently no consensus regarding the most effective type or long-term effectiveness of exercise to address the movement impairments seen in patients with PD, particularly those related to balance and gait.[3][4] Many current therapies are based on the notion of bypassing the disease-affected basal ganglia pathways, and instead using the frontal cortex to more deliberately perform movements with the help of external cues.[3] Common gait therapies for Parkinson's include using treadmill training, visual cues on the ground such as tape or laser-projected lines, rhythmic auditory cues, or segmentation of sequential movements.[3] Other commonly researched physical interventions for Parkinson's include mental rehearsal and visualization of movement prior to performing it, dance, and martial arts.[3][4] However, there is insufficient evidence to show significant differences between the effectiveness of any one of the aforementioned approaches over another.[3][4]
Recent research has shown promising effects through the use of Tai Chi, a form of balance-based martial arts, in managing and even reducing the loss of movement in patients with PD.[5][6] One study by Li and colleagues in 2012 found “improvement in maximum excursion with reduced deviation in movement,” “ability of the participants to adopt effective sway strategies (at the ankle or hip)”, and “marked increase in gait velocity… with significant increases in stride length” after patients with Parkinson's participated in a Tai Chi exercise program twice a week for 24 weeks.[5] A systematic review and meta-analysis by Ni and colleagues in 2014 reported that the variation of movements used in Tai Chi, including single-leg balancing, weight shifting and trunk rotation helps to train and improve overall balance and postural stability, especially when this exercise approach is combined with pharmacological interventions for optimal benefit.[6] Patient satisfaction with activity participation, as well as equipment and financial feasibility all play an important role in the the likelihood that an individual will adhere to an exercise program.[6] Given the adaptability of the movements and minimal costs and equipment associated with Tai Chi, it is an attractive and feasible option for the management of mild to moderate stages of PD.[6]
Client Characteristics[edit | edit source]
Mrs. A is a 67 year old female who was diagnosed with Parkinson's 6 years ago, for which she takes 250 mg t.i.d. of Levodopa. She is otherwise healthy, and up until recently, experienced only minimal symptoms associated with PD. Mrs. A has noticed increasing difficulty with gait and other movements of her extremities, as well as increased tremor in her hands over the last 3 months. Grocery shopping and cooking have been most difficult for Mrs. A, as well as cleaning around the house, especially when it involves reaching overhead or wiping motions. Mrs. A avoids wearing buttoned shirts or pants with zippers because of difficulties with these fine motor tasks. She is seeking physiotherapy from the author to help manage her progressing symptoms to maintain maximal function and independence.
Examination Findings[edit | edit source]
Subjectively, Mrs. A reported that she was experiencing increasing difficulty voluntarily moving her extremities, and had feelings of being “stuck and unsteady”, especially when she was in a hurry. She was a retired teacher who lived alone, and was completely independent with her daily activities and self-care, however daily tasks such as dressing and cooking were becoming much more difficult for Mrs. A in recent months. She expressed that her primary concern was not being able to care for herself as the disease continued to progress. She had also noticed recently that it was taking her longer to respond to questions or participate in conversation at the same speed as those she was conversing with. Mrs. A did not use any mobility aid, and did not have any investigations done prior to the assessment. She had also taken her medication two hours prior to attending her hour-long physiotherapy assessment.
Upon objective examination, she exhibited slight head forward posture with increased kyphosis of the thoracic spine, internally rotated shoulders, and stood in slight hip flexion. She was able to actively approach a more upright posture with normal spinal curvatures upon cueing, but was unable to maintain this position for more than five seconds. A resting tremor was observed in both hands, and bilateral intention tremor was also seen with finger-to-nose task. AROM testing showed decreased cervical and thoracic spine extension, limited shoulder abduction and flexion bilaterally, limited shoulder external rotation, on the left more than the right (patient is RHD), and decreased hip extension bilaterally. PROM was full for both upper and lower extremities, except for shoulder flexion bilaterally. Mild rigidity was present in the PROM testing of the trunk in rotation to both sides, and bilateral hips and knees; minor cogwheel rigidity was also observed during PROM testing in both wrists. Mrs. A demonstrated good strength of all major muscle groups, however hip flexion and extension were weaker on the left than on the right. Neurological testing was within normal limits.
Gait analysis revealed overall slow gait speed, decreased but present trunk rotation to both sides, minimal reciprocal arm swing bilaterally, and asymmetrical gait, with the left step length being shorter than the right. Push-off, heel strike, knee and hip range of motion were decreased bilaterally throughout the gait cycle, and right-sided trunk leaning was exhibited during left swing phase. Pill-rolling in the hands increased during gait, and there was initial freezing of about 3 seconds prior to starting, however Mrs. A did not exhibit freezing or festinating during gait. She was able to turn in both directions, with more difficulty turning to the left than the right.
The following are Mrs. A’s generalized results for the Movement Disorder Society - Unified Parkinson’s Disease Rating Scale (MDS-UPDRS).[7] Absolute scores not reported here.
- Link to Test (See pp. 2140-2170)
- Part I (Non-Motor Aspects of Experiences of Daily Living: Normal to slight impairments
- Part II (Motor Aspects of Experiences of Daily Living: Mild impairments
- Part III (Motor Examination): Mild impairments
- Part IV (Motor Complications): Slight to mild impairments
This is a disease-specific outcome measure used commonly in Parkinson's research. Parts I, II, and IV are self-reported, and Part III is a Motor Examination conducted by the clinician.[7] The test assesses the severity of both non-motor and motor impairments commonly seen in PD, as well as motor fluctuations experienced by the patient, where higher integer scores are indicative of greater severity of hallmark Parkinson's symptoms.[7] Studies have shown good reliability and validity for the use of MDS-UPDRS as a comprehensive evaluation of various aspects of PD.[8]
Mrs. A’s Hoehn and Yahr Rating was determined to be Stage 2.5, as indicated by her bilaterally occurring symptoms, and her ability to recover from the pull test in MDS-UPDRS within five steps. This rating scale, included as part of the MDS-UPDRS, gives a single numeric value to capture the severity and laterality of symptoms, balance, and gait impairments related to PD.[9]
Mrs. A completed the paper version of the Parkinson’s Disease Questionnaire-39 (PDQ-39), which was shown to have lower scores in four out of the eight main sections: mobility, ADLs, emotional well-being, and stigma. The others were reported to be less affected by Mrs. A’s condition. For the purposes of this case study, the four areas highlighted above will be the main focus for intervention. This disease-specific outcome measure, evaluating quality of life in patients affected by Parkinson's across eight different dimensions, shows similar, acceptable validity and high test-retest reliability (Cronbach’s alpha 0.72-0.95; test-retest 0.76-0.93 for paper version) in both the paper and electronic versions.[10][11]
As an adjunct to observational gait analysis and the Timed Up and Go (TUG) Test, the Freezing of Gait Questionnaire (FOGQ) was administered, on which Mrs. A scored 11/24.
- Link to Test (See Appendix B)
FOGQ is the only validated outcome measure currently available to evaluate freezing of gait (FOG) symptoms in Parkinson's and related conditions.[12] It is meant to be completed during the patient’s period of highest function within the “on” stage[13]. Because FOG occurs episodically and irregularly, patient self-report may be a more accurate representation of the extent to which FOG affects function.[12] As an example, Mrs. A did not exhibit much freezing during the observational gait analysis, however her FOGQ results suggest that she experiences these symptoms more often than the author was able to observe in the clinic.
The TUG test was performed to evaluate gait at a higher speed, as Mrs. A had reported difficulty ambulating, particularly when she was hurrying. Mrs. A last took her medication 2.5 hours prior to performing the TUG, towards the later portion of the ON-phase.[14]
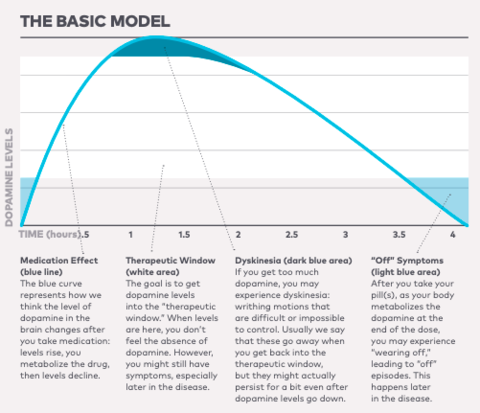
Source (https://www.parkinson.org/sites/default/files/attachments/MidStride.pdf, p. 14)[15]
Mrs. A’s TUG result was 13 seconds. The Timed-Up-And-Go (TUG) test has excellent inter-rater and test-retest reliability for differentiating between individuals with Parkinson's in “on” versus “off” phases, and when comparing individuals with Parkinson's in the “on” phase with control subjects, based on the time needed to complete the test.[16] The TUG has also been used as a predictor of fall risk in patients with PD, and was shown to accurately categorized more than 70% of a cohort study of over 2000 participants as fallers or non-fallers based on their TUG scores.[17] Nocera and colleagues, in 2013, proposed a cut score of 11.5 seconds, longer than which an individual with Parkinson's may be at increased risk of falls.[17] According to the cut score proposed here, Mrs. A’s score indicates that she is at a greater risk for falls. As Parkinson's is a progressive disease, it can be expected that this risk will only increase over time. The use of a gait aid during ambulation is to be considered in her treatment plan.
Clinical Impression[edit | edit source]
Mrs. A is a 67 y/o female who was diagnosed with Parkinson’s Disease six years ago. She self-referred to physiotherapy, and presents with recent worsening in bilateral resting and intention hand tremors, decreased static balance, and difficulty ambulating, secondary to her previously diagnosed PD. She is a suitable candidate for physiotherapy, and should tolerate treatment well due to her relatively high level of current function, as well as her desire to remain as independent as possible. Prognosis for physiotherapy will depend on the progression of her symptoms, however treatment will likely help in slowing the effects of disease progression on her daily function.
Problem List:[edit | edit source]
- Impaired balance due to poor posture (anteriorly displaced CoG)
- Abnormal gait pattern with asymmetrical step length and right-sided trunk leaning
- Increased risk of falls, as indicated by TUG score
- Decreased AROM in trunk, upper and lower extremities
- Head forward posture and increased kyphosis of thoracic spine
- Decreased strength in hip flexors and extensors
- Decreased fine motor control of hands and fingers
- Decreased ability and increased anxiety performing many ADLs secondary to loss of AROM, increased rigidity and loss of fine motor control
Intervention[edit | edit source]
Patient-centred treatment goals:[edit | edit source]
- Improve confidence and provide strategies for ambulating by 12 weeks, and re-assess using PDQ-39.
- Maintain AROM of upper extremities to help with grocery shopping and house cleaning in 24 weeks, to be re-assessed using goniometry.
- Reduce the risk of falls by 12 weeks, to be re-assessed using TUG test.
In discussion with Mrs. A regarding the prescription of a gait-aid to improve her safety when ambulating outside of her home, Mrs. A expressed not wanting to use a gait aid, as she did not feel as though her condition was at the point where she needed one. She said that she would rather dedicate her time to an exercise program to decrease her risk of falls than use a cane or walker.
To this end, Mrs. A was prescribed five 60-minute sessions of Tai Chi exercise per week for 24 weeks. These parameters were developed based on the 2012 study conducted by Li and colleagues,[5] in addition to the principles of neuroplasticity.[18] This type of activity is suitable for Mrs. A due to its adaptability and mindful style of practice. It can be practiced in standing or sitting, alone or in groups, and with or without an instructor. This leaves room for Mrs. A to be able to participate in the management of her condition and maintain her level of function as the disease progresses and her abilities change. Additional benefits that Mrs. A noted regarding Tai Chi was that it requires no additional equipment, and that it is very safe to practice alone due to its slow, controlled, but purposeful movements.[6] Further, many of the basic movement patterns used in Tai Chi mimic the ADLs and personal goals that Mrs. A had set out from the beginning.
The author chose not to prescribe Tai Chi every day because Mrs. A had voiced that the most attainable amount for her to do was five sessions per week; utilizing patient input while developing a treatment plan encourages greater adherence and participation. Further, with the stage of Parkinson's that she is currently in, Mrs A has the capability to do exercise more often.
The 2012 study by Li and team uses the prescription of two times per week for 24 weeks;[5] however, the principles of neuroplasticity suggest that a higher frequency of practice leads to stronger neural connections that are refined and reorganized with practice.[18] This suggests that implementing Tai Chi more often throughout the week could support better maintenance of the patient’s current function, in addition to creating a regular routine for Mrs. A. Further, the strongest predictor of learning a skill is the amount of times practiced, or frequency,[18] which was essentially the author’s main objective in the prescription of the Tai Chi exercise program. The primary goal was to retrain Mrs. A’s brain, through higher frequency practice, to bypass the basal ganglia in order to produce a desired movement. Tai Chi is a type of exercise that is formed through a group of basic movements,[5] so Mrs. A's week can be structured in a way where she attends an instructor-led session of Tai Chi, then applies the learning from this session into self-directed Tai Chi practice at home.
Lastly, there is evidence to support that the quality of mindfulness in Tai Chi encourages neuroplastic changes to occur within the brain. The movements of Tai Chi, in conjunction with the practice of mindfulness, promote neuroplasticity in the areas of the brain where attention, emotion, and self-awareness occur.[19] This suggests that Mrs. A may have been able to bypass the deficient basal ganglia via new neural connections created through neuroplasticity. In combination with the large movements associated with Tai Chi, these mechanisms may have helped Mrs. A to maintain her current level of function and improve the quality and amplitude of her movements.
Outcome[edit | edit source]
The Tai Chi program improved Mrs. A’s symptoms indirectly as measured by noticeable improvements in her TUG, UPDRS, and FOGQ scores during reassessment at 3 months.[3][5] Her TUG score at the 3 month re-assessment was 9 seconds, compared to 13 seconds at baseline (MCID = 3.5 seconds,[20] suggesting a lower risk of falls and improved balance and safety while ambulating.[17] There were noticeable improvements in UPDRS Part II (Motor Aspects of Experiences of Daily Living), and some performance improvements in specific tasks of Part III (Motor Examination); namely gait and freezing of gait. There was no appreciable change within Part I (Non-Motor Aspects of Experiences of Daily Living) or Part IV (Motor Complications) at the 3 month re-assessment. Her FOGQ score at 3 months was 9/24 (MCID = 1.4 points[3]). In accordance with her scores, Mrs. A subjectively reported feeling more sturdy while walking and less hesitation when accelerating while walking, such as when trying to catch the bus.
Mrs. A's confidence in performing ADLs such as wiping objects, grocery shopping and cooking improved as indicated by her PDQ-39 score. Her scores improved within the mobility, ADLs and emotional well-being categories (Mobility MCID = -1.5, ADL MCID = -0.7, emotional well-being MCID = 0.3)[21], but there was not a significant change within the stigma category. Mrs. A also subjectively reported that she was still having difficulty with tasks involving fine motor movements of the hands because of her tremor, which had not changed.
Mrs. A had an improved gait pattern, with increased gait speed, stride length, trunk rotation and bilateral arm swinging. It is inconclusive as to whether Tai Chi could have longer-term effects on gait due to lack of evidence in this area.[5][6]
Mrs. A’s posture had not changed since baseline. This is consistent with the lack of evidence to support direct effects of Tai Chi on postural changes.[6]
Discharge Plan:[edit | edit source]
Due to the progressive nature of this disease and lack of evidence in the literature, long-term effects of physiotherapy interventions are inconclusive. Therefore upon completing the 24 week Tai Chi program and achieving her goals, Mrs. A was discharged from physiotherapy care and educated regarding self-management of her condition. Mrs. A was referred to an occupational therapist by her family physician for future management of her ADL’s and for modifications that may be required in her home in the future. Mrs. A also received a referral to a speech-language pathologist so that she could work on her difficulties with speech initiation, as well as to manage any other speech impairments that may occur in the future.
Discussion[edit | edit source]
Mrs. A was a 67 y/o female diagnosed with Parkinson's six years ago, and her chief concerns that were targeted with physiotherapy treatment were increasing difficulty with gait and other movements of her lower extremities as well as several participation limitations, specifically grocery shopping, cooking and cleaning. Several outcome measures were used to track changes over time, including the TUG for balance and gait, the MDS-UPDRS to encompass all areas affected by PD, the FOGQ, and the PDQ-39 to evaluate Mrs. A’s perception of her quality of life. Mrs. A expressed a strong desire to remain functionally independent and was a suitable candidate for physiotherapy. She participated in a 24 week Tai Chi program, which lead to improvements in her balance, gait, freezing of gait symptoms, and quality of life. Significant changes in outcome measures were observed at reassessment after 3 months of intervention. Mrs. A was discharged upon completion of the Tai Chi program at 24 weeks, as she had achieved her physiotherapy goals; re-assessment of the above outcome measures showed maintenance of her progress at 6 months after initial assessment. She was given education regarding self-management of her condition, along with referrals to an occupational therapist and a speech-language pathologist. Mrs. A also reported that she would continue to participate in a community-based and self-directed Tai Chi program following discharge from physiotherapy.
The movement sequences associated with Tai Chi challenges individuals’ balance due to constant shifting of the base of support. For patients with PD, Tai Chi has been shown to have a beneficial short-term impact in maintaining movement, gait, and balance.[5][6] The use of mindfulness contributes to the promising evidence for improving quality of life in patients with PD.[22] Exercise is a well-supported therapy for Parkinson's management, however the most effective type of exercise and its effectiveness over time remains inconclusive.[3][4] Further research with larger sample sizes and longer duration of follow-up is still needed to determine the effect size of this intervention.
Self-Study Questions[edit | edit source]
- According to current evidence, which of the following motor aspects of a Parkinson’s patient would most likely be influenced by a Tai Chi exercise program?
- A: Rigidity
- B: Gait
- C: Tremor
- What is the proposed cut-off score for the TUG in patients with Parkinson's?
- A: 20.5
- B: 13
- C: 11.5
- D: 15
- What type of exercise has sufficient evidence to show that it is the best for patients with Parkinson’s?
- A: Gait training on a treadmill
- B: Dance
- C: Martial arts
- D: None of the above
Answers[edit | edit source]
- B
- C
- D
- ↑ 1.0 1.1 Opara JA, Brola W, Leonardi M, Blaszczyk B. Quality of life in Parkinson’s Disease. J Med Life 2012;5(4):375-381. PMID: 23346238
- ↑ 2.0 2.1 2.2 Wong SL, Gilmour H, Ramage-Morin PL. Parkinson’s disease: prevalence, diagnosis and impact. Health Reports 2014;25(11):10-14.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 Tomlinson CL, Patel S, Meek C, Herd CP, Clarke CE, Stowe R, et al. Physiotherapy versus placebo or no intervention in Parkinson’s disease. Cochrane Database of Systematic Reviews [Internet]. 2013 Sep 10 [cited 2019 May 4]. Available from: https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD002817.pub4/full
- ↑ 4.0 4.1 4.2 4.3 Schenkman M, Hall DA, Baron AE, Schwartz RS, Mettler P, Kohrt WM. Exercise for people in early- or mid-stage Parkinson disease: a 16-month randomized controlled trial. Phys Ther 2012;92(11):1394-1410. PMID: 22822237
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 5.7 Li F, Harmer P, Fitzgerald K, Eckstrom E, Stock R, Galver J, et al. Tai Chi and Postural Stability in Patients with Parkinson’s Disease. N Engl J Med 2012;366:511-519. Available from: 10.1056/NEJMoa1107911
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 Ni X, Liu S, Lu F, Shi X, Guo X. Efficacy and Safety of Tai Chi for Parkinson’s Disease: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. PLOS ONE 2014;9(6):e99377. Available from: https://doi.org/10.1371/journal.pone.0099377
- ↑ 7.0 7.1 7.2 Goetz CG, Tilley BC, Shaftman SR, Stebbins GT, Fahn S, Martinez-Martin P, et al. Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): Scale Presentation and Clinimetric Testing Results. Mov Disord 2008;23(15):2129-2170.
- ↑ Martinez-Martin, P. Instruments for holistic assessment of Parkinson’s disease. J Neural Transm 2013;120(4):559-564.
- ↑ European Parkinson’s Disease Association [Internet]. Kent UK: European Parkinson’s Disease Association; 2017. 3. Hoehn and Yahr Scale [cited 2019 May 2]; [about 1 screen]. Available from: https://www.epda.eu.com/about-parkinsons/symptoms/rating-scales/
- ↑ Morley D, Jenkinson C, Dummett S, Kelly L, Churchman DR, Dawson J. PND50 - The Parkinson’s disease questionnaire (Pdq-39) - Evaluating The Psychometric Properties of an Electronic Version. Value in Health 2015;18(3):A286. Available from: https://doi.org/10.1016/j.jval.2015.03.1670
- ↑ Hagell P, Nygren C. The 39 item Parkinson’s disease questionnaire (PDQ-39) revisited: implications for evidence based medicine. J Neurol Neurosurg Psychiatry 2007;78(11):1191-1198.
- ↑ 12.0 12.1 Giladi N, Shabtai H, Simon ES, Biran S, Tal J, Korczyn AD. Construction of freezing of gait questionnaire for patients with Parkinsonism. Parkinsonism Relat Disord 2000;6:165-170.
- ↑ Shirley Ryan Abilitylab. Freezing of Gait Questionnaire [Internet]. Chicago IL: Shirley Ryan Abilitylab; 2012 [updated 2012 Jul 25; cited 2019 April 30]. Available from: https://www.sralab.org/rehabilitation-measures/freezing-gait-questionnaire#parkinsons-disease
- ↑ Parkinson’s Foundation. Managing Parkinson's Mid-Stride. A Treatment Guide to Parkinson’s Disease [Internet]. 2018 [cited 2019 April 30]. p. 14. Available from: https://www.parkinson.org/sites/default/files/attachments/MidStride.pdf
- ↑ Parkinson’s Foundation. Visual representation of Levodopa effectiveness and dopamine levels over time in patients with PD. [Image on Internet]. 2018 [cited 2019 May 5]. p. 14. Available from: https://www.parkinson.org/sites/default/files/attachments/MidStride.pdf
- ↑ Morris S, Morris ME, Iansek R. Reliability of Measurements Obtained with the Timed “Up & Go” Test in People with Parkinson Disease. Phys Ther 2001;81:810-818.
- ↑ 17.0 17.1 17.2 Nocera JR, Stegemoller EL, Malaty IA, Okun MS, Marsiske, M, Hass, CJ. Using the Timed Up & Go Test in a Clinical Setting to Predict Falling in Parkinson’s Disease. Arch Phys Med Rehabil 2013;94:1300-1305.
- ↑ 18.0 18.1 18.2 Blumenfeld, H. Neuroanatomy through Clinical Cases 2nd ed. Massachusetts: Sinauer Associates Inc; 2010.
- ↑ Tang, Y., Holzel, B.K., Posner, M.I. The neuroscience of mindfulness meditation. Nature Reviews. Neuroscience. 2015. 16; 213-225.
- ↑ Shirley Ryan Abilitylab. Timed Up and Go [Internet]. Chicago IL: Shirley Ryan Abilitylab; 2013 [updated 2013 Nov 6; cited 2019 May 5]. Available from: https://www.sralab.org/rehabilitation-measures/timed-and-go
- ↑ Shirley Ryan Abilitylab. Parkinson’s Disease Questionnaire-39 [Internet]. Chicago IL: Shirley Ryan Abilitylab; 2014 [updated 2014 Jan 29; cited 2019 May 5]. Available from: https://www.sralab.org/rehabilitation-measures/parkinsons-disease-questionnaire-39
- ↑ Song R, Grabowski W, Park M, Osypiuk K, Vergara-Diaz GP, Bonato P, et al. The impact of Tai Chi and Qigong mind-body exercises on motor and non-motor function and quality of life in Parkinson's: A systematic review and meta-analysis. US National Library of Medicine National Institutes of Health 2017;41:3-13. Available from: 10.1016/j.parkreldis.2017.05.019