Emery Dreifuss Muscular Dystrophy: Difference between revisions
No edit summary |
No edit summary |
||
| Line 72: | Line 72: | ||
== Systemic Involvement == | == Systemic Involvement == | ||
Emery Dreifuss Muscular Dystrophy affects voluntary muscles, as well as the heart. Symptoms typically reveal themselves by the age of 10. Early symptoms include weakness and atrophy in a humero-peroneal distribution, that will eventually affect the scapular and pelvic girdle muscles.<ref name="GeneReviews" /> Contractures will occur, and can make arm, neck, ankle, and spine movements difficult, leading to to "toe-walking" and difficulty bending the elbows.<ref name="MDA" /><br> | |||
Cardiac conduction defecits and arrythmias may occur, resulting in dilated cardiac myopathy, poor exercise tolerance, bradycardia, syncope, congestive heart failure, and an increased risk of stroke and sudden death.<ref name="GHR" /><ref name="EJHG" /> | |||
Women who are genetic carriers for X-linked EDMD may also be at risk for cardiac problems, with the risk increasing with age. However, carriers tend not to present with muscle weakness or contractures.<ref name="MDA" /> <br> | |||
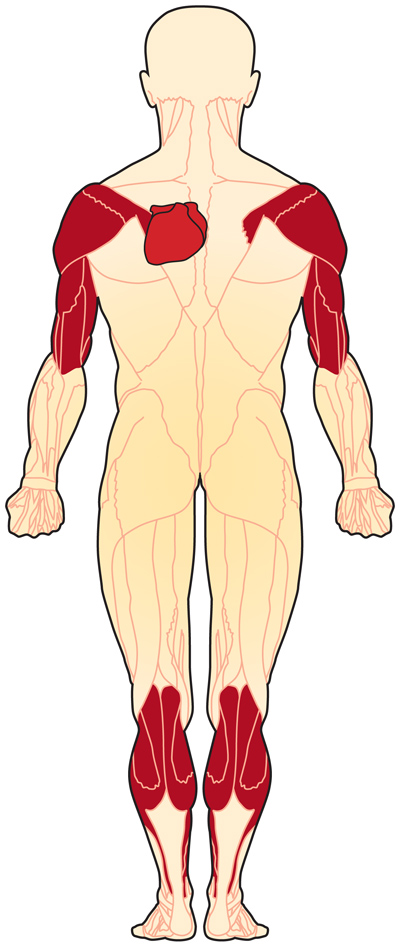
[[Image:Body_EDMD.jpg|Areas affected by Emery Dreifuss Muscular Dystrophy]] | |||
== Medical Management (current best evidence) == | == Medical Management (current best evidence) == | ||
Revision as of 21:00, 5 March 2014
Original Editors - Kori Ivanchak & Madelaine Nolan from Bellarmine University's Pathophysiology of Complex Patient Problems project.
Top Contributors - Madelaine Nolan, Rucha Gadgil, Kim Jackson, Elaine Lonnemann, Meaghan Rieke, Lucinda hampton, Kehinde Fatola, Shreya Pavaskar, Wendy Walker, Evan Thomas and WikiSysop
Definition/Description[edit | edit source]
add text here
Prevalence[edit | edit source]
There are approximately 250,000 individuals in the United States affected by some type of muscular dystrophy.[1]
The incidence of X-linked Emery Dreifuss Muscular Dystrophy is estimated to affect 1 in 100,000 people in the general population[2][3], and it is believed to be the third most common type of muscular dystrophy.[1] X-linked EDMD is fully expressed in males only; however, 10-20% of female carriers for X-linked EDMD will develop heart conduction defects and/or muscle weakness.[1]
The frequency of autosomal dominant EDMD is unknown. The autosomal recessive type of the disorder is very rare, with only a few reported cases worldwide.[3] The autosomal types of EDMD affect males and females equally. [1]
Characteristics/Clinical Presentation[edit | edit source]
add text here
Associated Co-morbidities
[edit | edit source]
Individuals with EDMD present with a wide variety of cardiac abnormalities including atrial standstill, atrial fibrillation, congestive heart failure, and cardioembolic stroke. The population is at high risk for devloping severe bradyarrhythmias which carries a risk of sudden death, and supraventricular tachyarrhythmias which are associated with a high risk for thromboembolic stroke. It is important to note that cardiac and muscular involvement have not been shown to be closesly related, with cases of severe cardiomyopathy occuring in patients with only mild muscular symptoms. It is possible for stroke to be the first clinical manifestation of EDMD.[4]
Medications[edit | edit source]
add text here
Diagnostic Tests/Lab Tests/Lab Values[edit | edit source]
Clinical Diagnosis[5]
The clinical diagnosis of EDMD is made based on the presence of the triad that characterizes it:
- Early Contractures
- Slowly progressive wasting and weakness of the humero-peroneal muscles
- Cardiac conduction defects and arrhythmias
Other Nonspecific Clinical Findings[5]
- Electromyogram (EMG) usually show myopathic features with normal nerve conduction studies. However, neuropathic patterns have been discovered in X-Linked EDMD and autosomal dominant EDMD.
- CT scan shows a diffuse pattern of involvement in muscles including the biceps, soleus, peroneal, external vasti, gluteus, and paravertebral muscles. Findings in the calf and posterior thigh have been reported in patients with Autosomal Dominant EDMD.
Nonspecific Laboratory Findings[5]
- Serum CK Concentration may be normal or moderately elevated up to 2-20x upper normal level. Increases are seen more often at the beginning of the disease as opposed to later stages.
- Muscle Histopathology shows nonspecific myopathic or dystrophic changes: variation in fiber size, increase in internal nuclei, increase in endomysial connective tissue, necrotic fibers. Electron Microscopy may show specific alterations in nuclear architecture. Muscle Biopsy is rarely performed for diagnostic purposes due to lack of specificity.
Immunodetection of Emerin (detected by immunofluorescence and/or by western blot): Absent in 95% of individuals with X-Linked EDMD. Normally expressed in individuals with Autosomal Dominant EDMD. Immunodetection of FHL1 (detected by immunofluorescence and/or by western blot): Absent or significantly decreased in individuals with FHL1-related X-Linked EDMD.
Genetic Testing
Genetic testing can determine the presence of particular defects that cause EDMD and can help predict the course of the disease, as well as help assess the risk of passing the disease on to the next generation.[6]
Etiology/Causes[edit | edit source]
add text here
Systemic Involvement[edit | edit source]
Emery Dreifuss Muscular Dystrophy affects voluntary muscles, as well as the heart. Symptoms typically reveal themselves by the age of 10. Early symptoms include weakness and atrophy in a humero-peroneal distribution, that will eventually affect the scapular and pelvic girdle muscles.[5] Contractures will occur, and can make arm, neck, ankle, and spine movements difficult, leading to to "toe-walking" and difficulty bending the elbows.[6]
Cardiac conduction defecits and arrythmias may occur, resulting in dilated cardiac myopathy, poor exercise tolerance, bradycardia, syncope, congestive heart failure, and an increased risk of stroke and sudden death.[3][2]
Women who are genetic carriers for X-linked EDMD may also be at risk for cardiac problems, with the risk increasing with age. However, carriers tend not to present with muscle weakness or contractures.[6]
Medical Management (current best evidence)[edit | edit source]
add text here
Physical Therapy Management (current best evidence)[edit | edit source]
add text here
Alternative/Holistic Management (current best evidence)[edit | edit source]
add text here
Differential Diagnosis[edit | edit source]
add text here
Case Reports/ Case Studies[edit | edit source]
add links to case studies here (case studies should be added on new pages using the case study template)
Resources
[edit | edit source]
add appropriate resources here
Recent Related Research (from Pubmed)[edit | edit source]
see tutorial on Adding PubMed Feed
Extension:RSS -- Error: Not a valid URL: addfeedhere|charset=UTF-8|short|max=10
References[edit | edit source]
see adding references tutorial.
- ↑ 1.0 1.1 1.2 1.3 Lammerding, J. Emery Dreifuss Muscular Dystrophy. Available at: http://www.rarediseases.org/rare-disease-information/rare-diseases/byID/590/viewFullReport. Accessed March 3, 2014. (accessed 3 March 2014).
- ↑ 2.0 2.1 Helbling-Leclerc A, Bonne G, Schwartz K. Emery Dreifuss Muscular Dystrophy. http://www.nature.com/ejhg/journal/v10/n3/full/5200744a.html (accessed 3 March 2014).
- ↑ 3.0 3.1 3.2 Emery Dreifuss Muscular Dystrophy. http://ghr.nlm.nih.gov/condition/emery-dreifuss-muscular-dystrophy (accessed 3 March 2014).
- ↑ Boriani G, Gallina M, Merlini L, et al. Clinical Relevance of Atrial Fibrillation/Flutter, Stroke, Pacemaker Implant, and Heart Failure in Emery-Dreifuss Muscular Dystrophy: A Long-Term Longitudinal Study . Stroke . 2003;34 (4 ):901–908. Available at: http://stroke.ahajournals.org/content/34/4/901.abstract.
- ↑ 5.0 5.1 5.2 5.3 Bonne G, Leturcq F, Yaou RB. Emery Dreifuss Muscular Dystrophy. http://www.ncbi.nlm.nih.gov/books/NBK1436/ (accessed 3 March 2014)
- ↑ 6.0 6.1 6.2 Hauptmann-Thanheuser. Emery Dreifuss Muscular Dystrophy: Diagnosis. http://mda.org/disease/emery-dreifuss-muscular-dystrophy/diagnosis (accessed 3 March 2014)