Acute Respiratory Distress Syndrome (ARDS)
Original Editors - Students from Glasgow Caledonian University's Cardiorespiratory Therapeutics Project.
Top Contributors - Geoff Boudreau, Matt Ross, Lucinda hampton, Kim Jackson, Adam Vallely Farrell, Chrysolite Jyothi Kommu, Hugh Collins, Mohit Chand, Vidya Acharya, Admin and Michelle Lee
Definition[edit | edit source]
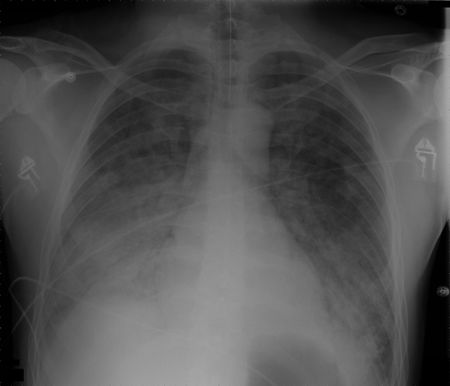
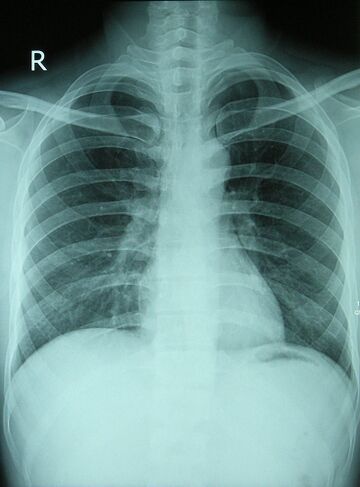
Acute respiratory distress syndrome (ARDS) [chest Xray R]
- A life-threatening condition of seriously ill patients, characterized by poor oxygenation, pulmonary infiltrates, and acuity of onset.
- On a microscopic level, the disorder is associated with capillary endothelial injury and diffuse alveolar damage.
- Carries a high mortality, and only a few effective therapeutic modalities exist to combat this condition.
- Once ARDS develops, patients usually have varying degrees of pulmonary artery vasoconstriction and, subsequently, may develop pulmonary hypertension.
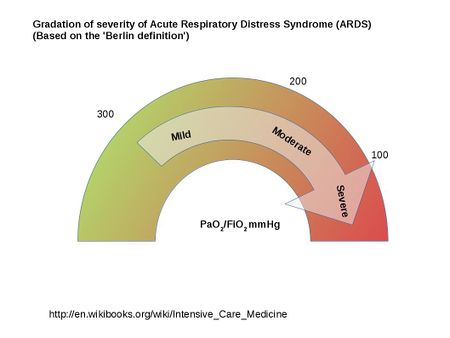
ARDS definition
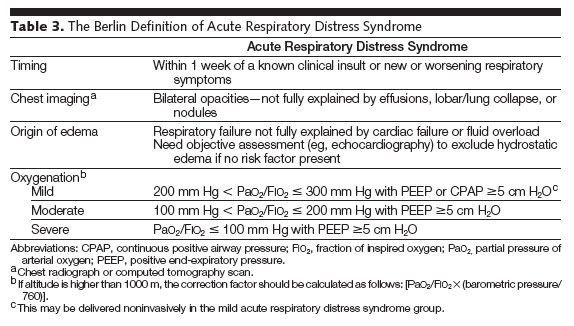
- An acute disorder that starts within 7 days of the inciting event and is characterized by bilateral lung infiltrates and severe progressive hypoxemia in the absence of any evidence of cardiogenic pulmonary edema.
- Defined by the patient's oxygen in arterial blood (PaO2) to the fraction of the oxygen in the inspired air (FiO2). These patients have a PaO2/FiO2 ratio of less than 300. The definition of ARDS was updated in 2012 and is called the Berlin definition[1].
Etiology[edit | edit source]
ARDS has many risk factors.
- Pulmonary infection e.g. the occurrence rate of ARDS with COVID-19 infection varies between 17% and 41%[2]
- Pulmonary aspiration
- Extra-pulmonary sources include sepsis, trauma, massive transfusion, drowning, drug overdose, fat embolism, inhalation of toxic fumes, and pancreatitis (these extra-thoracic illnesses and/or injuries trigger an inflammatory cascade culminating in pulmonary injury)[1].
Some risk factors for ARDS include:
- Advanced age
- Female gender
- Smoking
- Alcohol use
- Aortic vascular surgery
- Cardiovascular surgery
- Traumatic brain injury
Epidemiology[edit | edit source]
- Estimates of the incidence of ARDS in the United States range from 64.2 to 78.9 cases/100,000 person-years.
- Twenty-five percent of ARDS cases are initially classified as mild (1/3 will progress to severe or moderate) and 75% as moderate or severe.
- A literature review revealed a mortality decrease of 1.1% per year for the period 1994 through 2006.
- The mortality of ARDS is dependant on the severity of the disease, it is 27%, 32%, and 45% for mild, moderate, and severe disease, respectively[1].
- The prevalence of people suffering from ARDS differs greatly between geographical areas. Although the reason for the differences is unclear, some have speculated that it may stem from the differences in healthcare systems[3]. The ability to diagnose and to differentiate the secondary condition is required to properly record and treat the patient.
Pathological Process[edit | edit source]
ARDS represents a stereotypic response to various etiologies.
It progresses through different phases.
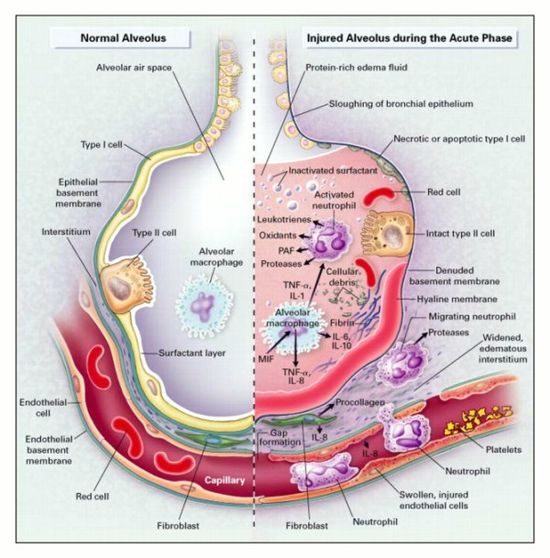
- First phase - Damage of the alveolo-capillary barrier leading to pulmonary oedema. The pulmonary epithelial and endothelial cellular damage is characterized by inflammation, apoptosis, necrosis, and increased alveolar-capillary permeability, which leads to the development of alveolar edema and proteinosis. There is bidirectional leakage of fluids and proteins into the alveolus and as well as surfactant proteins and alveolar cytokines into the plasma. The epithelial barrier becomes disrupted with a proliferation of type 2 alveolar cells leading to surfactant dysfunction. Alveolar edema, in turn, reduces gas exchange, leading to hypoxemia.
- Proliferative phase - characterized by improved lung function and healing.
- Final fibrotic phase - signaling the end of the acute disease process. Surfactant turnover is significantly increased and the fluid that lines the epithelium also highlights fibrosing alveolitis early in the course of lung damage.
- A hallmark of the pattern of injury seen in ARDS is that it is not uniform. Segments of the lung may be more severely affected, resulting in decreased regional lung compliance, which classically involves the bases more than the apices.
- This intrapulmonary differential in pathology results in a variant response to oxygenation strategies. While increased positive end-expiratory pressure (PEEP) may improve oxygen diffusion in affected alveoli, it may result in deleterious volutrauma and atelectrauma of adjacent unaffected alveoli.[1]
This 100 second video is a good introduction to ARDS.
Cellular Involvement in ARDS:
- Neutrophils: Most abundant in both the epithelial lining and alveolar histological specimens. Although the chemotaxic nature of neutrophils crossing the epithelium does not cause damage, their pro-inflammatory nature releases reactive oxygen species, cytokines and a number of inflammatory mediators which contribute to the basement membrane damage.
- Alveolar Macrophages: These are the most common cell type and with interstitial macrophages play an important role in defence. In ARDS patients there is a progressive increase in alveolar macrophage number.
- Epithelium: Contained within the alveolar epithelial tissue are the highly metabolically active type 2 alveolar cells. Damaged epithelium leads to dysfunctional surfactant.
Clinical Presentation and Assessment[edit | edit source]
The syndrome is characterized by:
- The development of dyspnea and hypoxemia, which progressively worsens within hours to days, frequently requiring mechanical ventilation and intensive care unit-level care.
- The history is directed at identifying the underlying cause which has precipitated the disease.
- When interviewing patients that are able to communicate, often they start to complain of mild dyspnea initially, but within 12-24 hours, the respiratory distress escalates, becoming severe and requiring mechanical ventilation to prevent hypoxia.
- The etiology may be obvious in the case of COVID 19, pneumonia or sepsis. In other cases, however, questioning the patient or relatives on recent exposures may also be paramount in identifying the causative agent.
The Physical Assessment may find.
- Acute Inflammatory Phase: Lasts 3 – 10 days and results in hypoxaemia and multi organ failure. Patients typically present with progressive dyspnoea, tachypnoea, cyanosis, hypoxic confusion and lung crepitations.
- Systemic signs (depending on the severity of illness) eg central or peripheral cyanosis as a result of hypoxemia, tachycardia, and altered mental status.
- Despite 100% oxygen, patients have low oxygen saturation. Chest auscultation usually reveals rales, especially bibasilar, but are often auscultated throughout the chest.[1]
- Healing, Proliferative Phase: During this phase, lung scarring and pneumothoracies are common.
Note that in both phases, secondary systemic and chest infections can occur.
Diagnostic Procedures[edit | edit source]
According to the National Heart, Lung and Blood Institute a diagnosis will be made via the examination of your medical history, physical exam and test results.[5]
Medical History[edit | edit source]
- History of heart failure
- Has the patient had any direct or indirect clinical risk factor for ARDS?
Physical Examination[edit | edit source]
- Added breath sounds on auscultation (e.g. crackling)
- Heart auscultation
- Cyanosis
Test Results[edit | edit source]
- Arterial blood gases
- Chest x-ray
- Blood Tests
- Sputum Culture
- CT Scan
- Heart Failure Examination
Management / Interventions[edit | edit source]
Unfortunately, no drug has been proven to be effective in preventing or managing ARDS.
The chief treatment strategy is supportive care and focuses on
- Reducing shunt fraction,
- Increasing oxygen delivery
- Decreasing oxygen consumption,
- Avoiding further injury.
Patients are mechanically ventilated, guarded against fluid overload with diuretics, and given nutritional support until evidence of improvement is observed.
The mode in which a patient is ventilated has an effect on lung recovery. Evidence suggests that some ventilatory strategies can exacerbate alveolar damage and perpetuate lung injury in the context of ARDS.
- Care is placed in preventing volutrauma (exposure to large tidal volumes), barotrauma (exposure to high plateau pressures), and atelectrauma (exposure to atelectasis).[1]
Possible Interventions for ARDS[edit | edit source]
NB Consider the risk vs reward of your intervention, particularly when the lungs are as fragile.
- Suctioning (Open / Closed)
- Ventilator Hyperinflation (VHI)
- Positioning (see below).
Ventilator Hyperinflation (VHI)[edit | edit source]
A systematic review by Anderson et al (2015) [6] considered that both manual and ventilator hyperinflation have similar affects on secretion clearance, pulmonary compliance, improve atelectasis and oxygenation without a detrimental risk to cardiovascular stability. However, a manometer should be used throughout the intervention with special attention paid to contraindications.
Due to the high levels of PEEP that are required to maintain lung recruitment for an ARDS patient, using the ventilator to deliver the hyperinflation is ideal. Also, VHI allows the constant monitoring and monitoring of airway pressures allowing the delivery to be titrated accordingly.
Prone Positioning[edit | edit source]
Placing the ARDS patient into prone will result in a significant increase in PaO2 for approximately 70% of the patients. By placing the patient into prone, there is an improvement in the recruitment of the dorsal aspect of the lung resulting in a more evenly distributed perfusion and improving V/Q matching.
Evidence has shown that prone positioning is beneficial particularly those who are severely hypoxaemic/severe ARDS, with reductions in ICU mortality without increased airway complications.
- Explain to the patient, with reassurance, that they will be safe and obtain consent if they are able to communicate.
- Close eyes and protect with gel or pad.
- Place the patient's palms against their thighs, thumbs upwards, elbows straight and shoulders neutral.
- Slide the patient to the edge using a Sliding sheet.
- Roll patient into the lateral position using the underneath sheet.
- Roll patient into prone.
- 'Swimmer's position' - elbow in which the head is semi-rotated should be flexed to no more than 90° to avoid ulnar nerve stretch, and the other arm internally rotated by the side.
- Ensure that women's breasts or men's genitals are not compressed.
- Place two pillows under each shin to prevent peroneal nerve stretch, positioning them to avoid knee and toe pressure from mattress. [7]
Mechanical Ventilation[edit | edit source]
As a result of the acute onset of hypoxaemic respiratory failure with an increased work of breathing, mechanical ventilation is commonplace. The pathophysiology of ARDS, and in particular the fibrosing aspect means that the method and delivery of support requires careful consideration to ensure that ventilator induced lung injury (VLII) does not occur[8]
In the ARDS patient, the PEEP and tidal volume has to be reduced to reflect the available parts of the lung that are available for aeration. If not, this can lead to an over stretching of the lung, known as volutrauma due to decreased lung compliance. Repeated open and closing of the alveoli during tidal inflation can also cause injury to the lung tissue, known as alectotrauma. If the mechanical support is not titrated to the patient, both alectotrauma and volutrauma can increased the cytokines within the lung tissue which then may enter the systemic circulation and potentially lead to multiple organ failure[8]
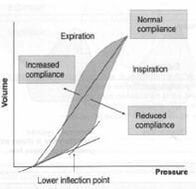
Although perhaps oversimplified, the graph below attempts to highlight the optimal area for ventilation of the ARDS patient. The lower inflection point (LIP) is thought to be the pressure at which lung tissue is recruited. The upper inflection point (UIP) is the approximate pressure at which the alveoli become over distended which can potentially lead to damage. Therefore, the most optimum position to ventilate would be between the two inflection points, on the steepest most complicant aspect of the curve[9].
Inverse Ratio Ventilation (IRV)[edit | edit source]
This mode of ventilation requires the alteration in the normal inspiratory / expiratory ratio of 1:2 to either 1:1 or 2:1. While this mode of ventilation aims to increase the aeration and collateral ventilation of the poorly compliant lung, decreased expiration time may result in an increase in the PCO2. However, if hypercapnic acidosis occurs slowly then the intracellular acidosis will be well compensated, however any form of hypercapnia should be avoided in this with or those at risk of developing raised intracranial pressures. This is known as permissive hypercapnia.
A recent although small study by Kotani et al (2016) [10]showed that IRV provided acceptable gas exchange without complications for 13 people with ARDS.
Airway Pressure Release Ventilation (APRV)[edit | edit source]
This form of mechanical ventilation involves the delivery of CPAP with an intermittent release phase. Delivering CPAP over long periods is thought to maintain adequate lung volume and alveolar recruitment where the release is designed to aid the removal of CO2. Inspiration starts from a more beneficial pressure-volume relationship thus helping oxygenation [11].
High Frequency Oscillation (HFO)[edit | edit source]
With HFO, a continuous distending pressure (CDP) is set, and small tidal volumes are delivered at high frequencies with an oscillatory pump. Like the others previously described, this is another form of lung protective ventilation with constant lung recruitment. While a systematic review by Sud et al (2010)[13] found that HFO may improve survival and is unlikely to cause harm, only 8 studies were analysed with few patients and wide confidence intervals. There were also no reports of blinding outcome assessors putting trials at risk of bias[13].
Extracorporeal Membrane Oxygenation (ECMO)[edit | edit source]
ECMO can be used for those patients who have severe respiratory failure and is used when conventional therapy cannot maintain adequate oxygenation. ECMO can fully replace the function of the lungs, however due to various complications there needs to be consideration of the risk vs benefit.
ECMO consists of an extracorporeal blood circuit formed via an oxygenator and a pump. Two vascular accesses are established, one removes the venous blood, and one infuses the oxygenated blood. After the blood has been removed from a major vein, it is pumped through a circuit that contains an oxygenator which oxygenates the blood and removes the carbon dioxide. The blood is then returned via the other cannula[15].
Inhaled Nitric Oxide[edit | edit source]
Not only is nitric oxide an endothelium-derived smooth muscle relaxant, it also assist with neurotransmission, host defence, platelet aggregation and bronchodilation. Inhaled nitric oxide may delivered continuously or using intermittent inspiratory injection and a rise in PaO2 of 20% is considered a positive response. 40% - 70% of ARDS patients have improved oxygenation following inhaled nitric oxide and is only commonly used in patients with severe hypoxemia as a temporary rescue. [8]
Role of the Physiotherapist[edit | edit source]
Input from physiotherapy is often limited and minimal often due to the need for high PEEP and high oxygen requirements. As it is interstitial pathology secretions are not generally a problem. Treatment may consist of positioning only, e.g. prone lying to optimise gas exchange. Caution is needed with hands-on techniques as you want to ensure you do not de-recruit lung units losing the splinting effect of the ventilator PEEP. If secretions become an issue, ensure adequate humidification along with other techniques to improve sputum clearance. Minimal suctioning via ETT. A state-of-art review suggests avoiding repeated airway clearance in infants and children with the acute pulmonary disease[17].
Conclusions[edit | edit source]
- Even though many risk factors for ARDS are known, there is no way of preventing ARDS.
- Careful management of fluids in high-risk patients can be helpful.
- Steps should be taken to prevent aspiration by keeping the head of the bed elevated before feeding.
- Lung protective mechanical ventilation strategy in patients without ARDS who are high risk would help prevent ARDS[1].
Resources[edit | edit source]
- Cochrane Review: Recruitment manoeuvres for adults with acute respiratory distress syndrome receiving mechanical ventilation
- Cochrane Review: Pressure-controlled versus volume-controlled ventilation for acute respiratory failure due to acute lung injury (ALI) or acute respiratory distress syndrome (ARDS)
- Cochrane Review: High versus low positive end-expiratory pressure (PEEP) levels for mechanically ventilated adult patients with acute lung injury and acute respiratory distress syndrome
- Cochrane Review: Extracorporeal membrane oxygenation for critically ill adults
- Acute Respiratory Distress Syndrome. The Berlin Definition
- ARDSNet: Mechanical Ventilation Protocol Summary
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Acute Respiratory Distress Syndrome (ARDS) Authors :Matthew Diamond1; Hector L. Peniston Feliciano2; Devang Sanghavi3; Sidharth Mahapatra4. Available from:https://www.ncbi.nlm.nih.gov/books/NBK436002/?report=printable (last accessed 28.6.20)
- ↑ Villar J, Confalonieri M, Pastores SM, Meduri GU. Rationale for prolonged corticosteroid treatment in the acute respiratory distress syndrome caused by coronavirus disease 2019. Critical Care Explorations. 2020 Apr;2(4).Available from:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7188431/ (last accessed 28.6.2020)
- ↑ Walkey, A., Summer, R., Vu. H., Alkana, P. Acute respiratory distress syndrome: epidemiology and management approaches. Clinical Epidemiology 2012; 4: 159-169
- ↑ USMLEQA formerly USMLEFastTrack. Acute Respiratory Distress Syndrome (ARDS). Available from: https://www.youtube.com/watch?v=IOj-4oH9ROY&t=7s [last accessed 31/3/2024]
- ↑ National Heart, Lung and Blood Institute (2018). ARDS. Available at: https://www.nhlbi.nih.gov/health-topics/ards [Accessed 25th July 2018].
- ↑ Anderson, A., Alexanders, J., Sinani, C., Hayes, S. and Fogarty, M., 2015. Effects of ventilator vs manual hyperinflation in adults receiving mechanical ventilation: a systematic review of randomised clinical trials. Physiotherapy, 101(2), pp.103-110.
- ↑ IntensiveCareUnit Ashford-Kent. Prone Positioning - Natalie Matthews. Available from: https://www.youtube.com/watch?v=iMKybCBUzqM [last accessed 31/3/2024]
- ↑ 8.0 8.1 8.2 Bernsten, A. and Soni. S. (2014). OH's Intensive Care Manual. 7th Edition. Elsevier LTD.
- ↑ Moloney, E.D. and Griffiths, M.J.D., 2004. Protective ventilation of patients with acute respiratory distress syndrome. British Journal of Anaesthesia, 92(2), pp.261-270.
- ↑ Kotani, T., Katayama, S., Fukuda, S., Miyazaki, Y. and Sato, Y., 2016. Pressure-controlled inverse ratio ventilation as a rescue therapy for severe acute respiratory distress syndrome. SpringerPlus, 5(1), p.716.
- ↑ Daoud, E.G., 2007. Airway pressure release ventilation. Annals of thoracic medicine, 2(4), p.176.
- ↑ Little Criticos,Airway Pressure Release Ventilation (APRV), Mechanical Ventilation
- ↑ 13.0 13.1 Sud, S., Sud, M., Friedrich, J.O., Meade, M.O., Ferguson, N.D., Wunsch, H. and Adhikari, N.K., 2010. High frequency oscillation in patients with acute lung injury and acute respiratory distress syndrome (ARDS): systematic review and meta-analysis. Bmj, 340, p.c2327.
- ↑ Baxter Healthcare - U.S. THE VEST airway clearance system | 3D Animation Video. Available from: https://www.youtube.com/watch?v=v4nh68K1184 [last accessed 31/3/2024]
- ↑ Aokage, T., Palmér, K., Ichiba, S. and Takeda, S., 2015. Extracorporeal membrane oxygenation for acute respiratory distress syndrome. Journal of intensive care, 3(1), p.17.
- ↑ CARDIOLOGIST CHANNELS.ECMO ( extracorporeal membrane oxygenation) BASIC CONCEPT FOR CATHETER. Available from: https://www.youtube.com/watch?v=EvMAnCiDk8s [last accessed 31/3/2024]
- ↑ Morrow BM. Airway clearance therapy in acute paediatric respiratory illness: A state-of-the-art review. South African Journal of Physiotherapy. 2019 Jun 25;75(1):12.