Abdominal Aortic Aneurysm
Top Contributors - Clinton Morris, Justin Rosinski, Elaine Lonnemann, Evan Thomas, Kim Jackson, Lucinda hampton, WikiSysop, David Olukayode and Admin
Definition/Description[edit | edit source]
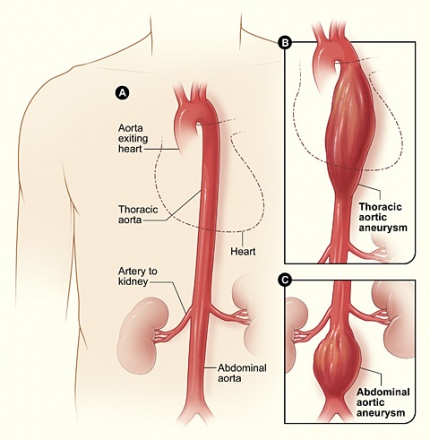
An aneurysm is defined as an increase in the diameter of a vessel compared to normal segments by 50% or more.[1] This occurs as a result of localized weakening of the arterial wall, and must include all 3 layers (intima, media, and adventitia) of the arterial wall to be considered a true aneurysm.[1] Aneurysms are most common in the aorta, particularly the abdominal and thoracic regions, but may also affect any part of the vascular system, including lesser arteries and veins.[2]
Although an aneurysm may occur in any artery, they are most common in the abdominal and thoracic regions.[1] The abdominal aorta begins at the aortic hiatus in the diaphragm (approximately T12 vertebral level), and ends at approximately the L4 vertebral level, where it splits into the right and left common iliac arteries.[3] The abdominal aorta is approximately 13 cm in length.[3] In the abdominal aorta, the most common location of an AAA is just below the renal arteries.[1] The most common complications arising from an abdominal aortic aneurysm (AAA) include pain from ischemia, thromboembolism, spontaneous dissection, and rupture of the aneurysm leading to hemorrhage and possible death.[1]
Prevalence[edit | edit source]
Abdominal aortic aneurysms account for three fourths of all aortic aneurysms, and have been reported to occur four times as often as a thoracic aortic aneurysm.[1][4] Occurrence of this condition is reported as 0.5-3.2% of the general population according to the Merck Manual ranging from 1.0-14.2% in men, and 0.2-6.4% in women.[1][5] This study found geographic differences, Australia having the highest incidence, followed by America, Europe, and Asia.[5] AAA’s are much more common in men, occurring up to four to six times more often.[1][6] The most typical location of an AAA is below the renal arteries (80%), although they may also occur lower, involving the iliac arteries.[1][4][6] A majority of AAA’s are fusiform, though some are saccular.[1] AAA’s are reported to be the 14th leading cause of death in the United States, causing 4500 deaths directly, with an additional 1400 deaths as a result of complications during surgical repair to prevent rupture.[6] It is also reported that over 45,000 surgeries are performed each year in an effort to prevent the rupture of an AAA.[6]
Pathogenesis[edit | edit source]
Only around 5% of all AAAs, known as inflammatory AAAs, have historically been thought to have a significant pathogenic role due to inflammation.[7] Inflammation, however, appears to play a significant role in the onset and progression of the majority of AAAs, even those that are not often classified as inflammatory AAAs, according to the majority of studies conducted over the past ten years. As a result of the discharge of a variety of proteolytic enzymes, such as matrix metalloproteinases and cysteine proteases, oxidation-derived free radicals, cytokines, and related products, chronic aortic inflammation is thought to cause the destruction of the aortic media and to vascular smooth muscle cell apoptosis and dysfunction.[8] A small number of studies suggest that immunosuppression encourages the progression and rupture of human AAA and cysteine proteases, oxidation-derived free radicals, cytokines, and related products, in contrast to the strong evidence implicating inflammation in AAA development and progression.[9][10] These results provide credence to the idea that inflammation connected to AAA is a reaction to the AAA rather than its root cause. Although there are various cautions to be taken into account, clinical trials focusing on different components of inflammation, such as cytotoxic T cell or cytokine inhibition, may provide a more accurate response as to whether chronic inflammation is a primary pathogenic mechanism behind AAA progression.[11]
Palpation Characteristics/Clinical Presentation[edit | edit source]
A large majority of abdominal aortic aneurysms are asymptomatic, and are often found incidentally as a result of other diagnostic imaging and procedures such as ultrasonography or magnetic resonance imaging.[1][6][4] Occult aneurysms may present with symptoms related to the underlying cause of the disease, such as extremity pain due to emboli, fever, malaise, etc.[1] Many AAA’s will remain asymptomatic until rupture.[6] When symptoms are produced, they typically include deep, boring pain in the abdomen, pain and tenderness to palpation, and prominent pulsating mass (greater than 3-4cm) in the abdomen.[6][1][4] Aggarwal et al. reports that symptomatic AAA’s are also more susceptible to rupturing.[6]
A ruptured aneurysm, if it does not cause sudden death (4-5% of all sudden deaths), will present with shooting pain in the abdomen or low back, pulsatile mass in the abdomen, tachycardia and severe hypotension.[6][1] Of patients who have a ruptured AAA, half will survive to reach the hospital, and of those, only 50% will survive the surgery to repair the artery.[6]
Associated Co-Morbidities[edit | edit source]
Smoking, or a history of smoking, is considered the strongest risk factor for abdominal aortic aneurysms.[1] Growth of the diameter of the aneurysm is correlated with highly with smoking, and a rapidly increasing diameter (>0.5 cm in 6 months) is associated with a higher risk of rupture.[6]
- Other associated risk factors include hypertension, atherosclerosis, coronary artery disease, older age (beginning at 55, with increasing prevalence at 70-80), family history (either of AAA, or for surgical intervention in a 1st degree relative), race (whites > blacks), and male sex.[1][6]
- For women, age is not considered a risk factor for up to 10 years later than men.[6]
- Those with Marfan’s syndrome and Ehlers-Danlos syndrome may also be at an increased risk of developing AAA’s.[12]
- High salt intake was independently associated with increased aortic diameter, although it is not known if reduction of salt intake would reduce this risk.[13]
- AAA’s have also been found to be less common in those with diabetes.[6]
- Patients who have diabetes mellitus and/or peripheral vascular disease are reported to have slower growth rates.[6][14]
Medications[edit | edit source]
Medications typically associated with the treatment of an abdominal aortic aneurysm, and/or associated co-morbidities listed above, include statins, angiotensin-converting enzyme inhibitors (ACE-inhibitors), angiotensin II receptor blockers, beta-blockers, antibiotic agents, and anti-inflammatory agents.[15][6] It is also important to consider poly-pharmacy effects in this population as older age is highly associated with developing AAA’s. Additionally, several newer therapies such as genetic modification and other forms of molecular modification therapies have been proposed.[15] Strategies targeting inflammatory mediators such as mast cells and macrophages, and therapies limiting the expression of targeted genes using synthetic gene “decoys” are examples of the alternative treatments currently being researched.[15]
Diagnostic Tests/Lab Values[edit | edit source]
As mentioned previously, diagnosis of an abdominal aortic aneurysm is often incidental, as a result of other diagnostic procedures.[1] AAA’s may be picked up by physical examination of the abdomen, where the clinician palpates the abdomen for an enlarged, pulsating mass greater than 3 cm.[1][6] There is some debate on the normal pulse width, with some considering 2.5-4.0 cm to be normal, while other sources say the width ought to be no greater 3 cm.[4] The average pulse width is reported as 2.5 cm or approximately 1.2 inches.[4] Additionally, palpation may be difficult to accurately perform in patients with abdominal distension or excessive adipose tissue.[6] The diameter of the AAA is also associated with risk of rupture.[1] The online Merck Manual provides a chart detailing the association between diameter and risk of rupture.[16]
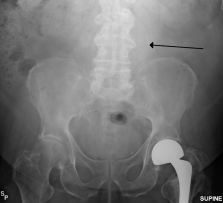
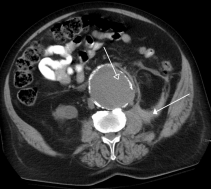
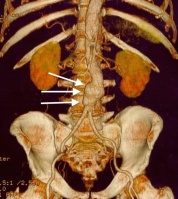
Other diagnostic procedures include ultrasound of the abdomen, CT with or without angiography, and magnetic resonance imaging with or without angiography.[1][6] Traditional radiographs can pick up on calcification of the arterial wall (although this is not always present).[1][6] Ultrasound is the diagnostic imaging technique of choice for screening due to low cost and quick results, as well as exceptional sensitivity and specificity (95-100% and close to 100% respectively).[6] Although ultrasound is preferred, the use of CT scanning may be used to better determine the size and shape of the aneurysm, and also to monitor the progression of an AAA in the long term.[1][6] MRI is not typically done specifically for the diagnosis of AAA’s due to exceptional cost and availability.[1][6]
Recently, there has been a push to screen more individuals considered to be at high risk for AAA’s using transthoracic echocardiography (TTE), which is a form of ultrasound.[17] Majeed et al. looked at the data from over 10,000 patients considered to be at an elevated risk of AAA and found that a significant portion of these patients were subsequently diagnosed with increased diameter of the abdominal aorta, indicating that increased screening high risk individuals on a regular basis may be indicated for better management of AAA’s and subsequent rupture.[17]
Laboratory values are not typically used in the diagnosis of a AAA, but it is common for tests to be run prior to surgery, including CBC, electrolytes, BUN, creatinine, PT, PTT, blood type and cross-matching.[1] There are no specific lab values to determine risk of, or the presence of AAA’s.[18][19][20][21]
Etiology/Causes[edit | edit source]
According to the Merck Manual, the most common cause of abdominal aortic aneurysms is weakening of the arterial wall, usually due to atherosclerotic processes.[1] Other causes include trauma (particularly with lifting), vasculitis, congenital vascular disease, cystic medial necrosis, and post-surgical anastomotic disruption.[1][4] Gene defects on chromosomes 11 and 15 have also been found to be associated with the development of aneurysms.[2] Less common causes include fungal and bacterial infections, causing infected or mycotic aneurysms.[1] Diseases such as HIV may place individuals at an increased risk of bacterial and fungal infection due to compromised immune systems.[2]
Systemic Involvement[edit | edit source]
Circulatory system
Increased aortic swelling causes blood to pool and swish around in the distended area, decreasing the efficiency of the blood to be pumped to the lower extremities, decreasing distal pulses (i.e dorsalis pedis). This ineffective delivery of blood may cause a decrease in temperature in the lower extremities and a difference in diastolic blood pressure between arms.[4][22]
Neurologic
Increased swelling of the abdominal aorta can cause increased pressure on the surrounding structures producing potential numbness in the lower extremities and pain in the abdomen, lower back, neck, and shoulder.[4][22]
Integumentary
Often times, individuals with AAA will under go surgery that results in an incision line that will need to be monitored for infection.[22]
Respiratory
After an AAA surgical repair, incisional pain and post surgical precautions hinder the function of abdominal muscle to produce an effective huffing/coughing along with completing full inspiration, predisposing a patient to increased risk of pulmonary complications.[22]
Medical Management[edit | edit source]
“Many approaches to treating aortic aneurysms exist, and a wide variety of devices are available. There is still no clear evidence that support one specific treatment regimen over another and, therefore, specific practice patterns vary between institutions.”[23]
Listed below are two surgical managements for patients with a large AAA (diameter of 5.5 cm or greater) and are asymptomatic:[6]
- Open Surgical Repair (removal of aneurysm)-An aggressive invasive procedure where the surgeon will make an incision down the midline of the patients’ stomach to reach the aneurysm site. A clamp will be place above and below the aneurysm to cut off blood flow in order for the aneurysm to be removed. The surgeon will then suture in new prosthetic graft material to replace the removed tissue. After the graft is in place, the clamps will be removed to restore circulation. Blood flow circulation will be monitored before incision site is closed.[22][24]
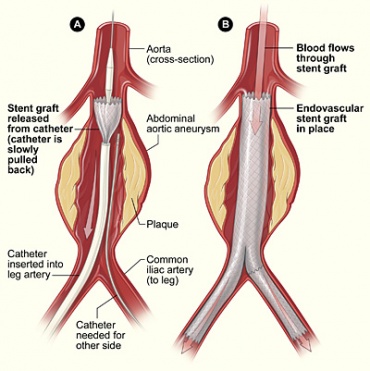
- Endovascular repair (sent grafts, aneurysm kept intact)-A less invasive, more widely accepted, and frequently practiced procedure, that is starting to become the gold standard for these surgeries. The surgeon will make a small incision into the groin area to find the femoral artery. A catheter balloon bladder is guided up to femoral artery via X-Ray to the site of the aneurysm location. Once in the desired location, the surgeon will open the stent and fix it to the wall of the aorta with sutures, leaving the aneurysm intact to shrink and grow around the stent. Blood flow is monitored after the procedure.[6][24][25]
Below are some conservative treatments that maybe more suitable for patients that are classified as having small to medium size aneurysms (<5.5cm determiner).[6]
- Beta Blockers- There is limited evidence on the use of beta blockers, but this invention has been shown to reduce the expansion rate of AAA’s especially in patients that have hypertension.[6]
- Antibiotics- Have been shown to reduce inflammation in AAA’s related to possible involvement of Chlamydia pneumonia.[6]
- Waiting/monitoring- Is usually for patients that have AAA diameters ranging from 3.0-4.0cms which should be monitored with medical imaging every two to three years. If the AAA increases in size to 4.0-5.4, that patient should be screened using medical imaging ever six to 12 months.[6]
Physical Therapy Management[edit | edit source]
There is currently no conservative management to affect the progression of an AAA but a physical therapist can provide education on risk factor reduction, cessation of smoking to reduce the detrimental effects of smoking, screening for signs/symptoms, and public education which has improved awareness of the disease.[6][1][26]
Cardiovascular fitness exercise (treadmill, cycle ergometer, stair climbing, elliptical training, and rowing 3 times weekly for 45 minutes at a target intensity of 60% of HR reserve initially and working up to 80% as tolerated) and resistance exercise have been found to be safe in improving functional status, potentially reducing cardiovascular risk factors, and showing no increased rate of AAA diameter progression in patients that have an AAA diameter of <5.5cm.[26]
Pre-operative physical therapy has also shown promising effects to decrease post AAA surgery complication risks, but is still unclear why and requires further research. Physical therapy with post surgical patients should focus on incisional site inspection and bronchial hygiene techniques to decrease incidence of pulmonary complications.[22][28]
Case Reports[edit | edit source]
- Mechelli F, Preboski Z, Boissonnault W. Differential Diagnosis of a Patient Referred to Physical Therapy With Low Back Pain: Abdominal Aortic Aneurysm. J Orthop Sports Phys Ther. 2008;38(9):551-557.
- Rodeghero J, Denninger T, Ross M. Abdominal Pain in Physical Therapy Practice: 3 Patient Cases. J Orthop Sports Phys Ther. 2013;43(2):44-53.
- Kim N, Kang S, Park S. Coexistence of expanding abdominal aortic aneurysm and aggravated intervertebral disc extrusion -a case report-. Korean Journal of Anesthesiology. 2013;65(4):345.
- Baskaran D, Ashraf N, Ahmad A, Menon J. Inflammatory abdominal aortic aneurysm: a persistent painful hip. Case Reports. 2013;2013(sep13 1):bcr2013009150-bcr2013009150.
- Kawatani Y, Nakamura Y, Hayashi Y, Taneichi T, Ito Y, Kurobe H et al. A Case Report on the Successful Treatment ofStreptococcus pneumoniae-Induced Infectious Abdominal Aortic Aneurysm Initially Presenting with Meningitis. Case Reports in Surgery. 2015;2015:1-6.
Resources[edit | edit source]
References[edit | edit source]
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 1.20 1.21 1.22 1.23 1.24 1.25 1.26 1.27 Porter RS, Kaplan JL. The Merck manual of diagnosis and therapy. Whitehouse Station, NJ: Merck Sharp & Dohme Corp.; 2011.
- ↑ 2.0 2.1 2.2 Goodman CC, Fuller KS. Pathology: implications for the physical therapist. St. Louis, MO: Saunders/Elsevier; 2009.
- ↑ 3.0 3.1 Moore KL, Agur AMR, Dalley AF, Moore KL. Essential clinical anatomy. Baltimore, MD: Lippincott Williams & Wilkins; 2015.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 Goodman CC, Snyder TEK. Differential diagnosis for physical therapists: screening for referral. St. Louis, MO: Saunders/Elsevier; 2013.
- ↑ 5.0 5.1 Li X, Zhao G, Zhang J, Duan Z, Xin S. Prevalence and Trends of the Abdominal Aortic Aneurysms Epidemic in General Population - A Meta-Analysis. Plos ONE [serial on the Internet]. (2013, Dec), [cited March 24, 2016]; 8(12): 1-11. Available from: Academic Search Complete.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 6.13 6.14 6.15 6.16 6.17 6.18 6.19 6.20 6.21 6.22 6.23 6.24 6.25 6.26 6.27 6.28 6.29 Aggarwal S, Qamar A, Sharma V, Sharma A. Abdominal aortic aneurysm: A comprehensive review. Experimental And Clinical Cardiology [serial on the Internet]. (2011, 2011 Spring), [cited March 24, 2016]; 16(1): 11-15. Available from: MEDLINE.
- ↑ Walker DI, Bloor K, Williams G, Gillie I. Inflammatory aneurysms of the abdominal aorta. Br J Surg. 1972;59(7):609-614.
- ↑ Golledge J. Abdominal aortic aneurysm: update on pathogenesis and medical treatments. Nat Rev Cardiol. 2019;16(4):225-242. doi: 10.1038/s41569-018-0114-9.
- ↑ Lindeman JHN, Rabelink TJ, Van Bockel JH. Immunosuppression and the abdominal aortic aneurysm: Doctor Jekyll or Mister Hyde? Circulation. 2011;124(4):463-465
- ↑ Tajima Y, et al. Oral steroid use and abdominal aortic aneurysm expansion — positive association. Circ J. 2017;81(12):1774-1782.
- ↑ Golledge J, Norman PE, Murphy MP, Dalman RL. Challenges and opportunities in limiting abdominal aortic aneurysm growth. J Vasc Surg. 2016;65(1):225-233.
- ↑ Aortic Aneurysm Fact Sheet [Internet]. Centers for Disease Control and Prevention. Centers for Disease Control and Prevention; 2013 [cited 2016Mar24]. Retrieved from: http://www.cdc.gov/dhdsp/data_statistics/fact_sheets/fs_aortic_aneurysm.htm
- ↑ Golledge J, Hankey G, Yeap B, Almeida O, Flicker L, Norman P. Reported High Salt Intake Is Associated with Increased Prevalence of Abdominal Aortic Aneurysm and Larger Aortic Diameter in Older Men. Plos ONE [serial on the Internet]. (2014, July), [cited March 24, 2016]; 9(7): 1-6. Available from: Academic Search Complete.
- ↑ Pixabay.com https://pixabay.com/en/smoking-smoke-cigarette-man-1026556/ Accessed on April 9, 2016
- ↑ 15.0 15.1 15.2 Miyake T, Morishita R. Pharmacological treatment of abdominal aortic aneurysm. Cardiovascular Research [Internet]. (2009, May 19) [cited March 24, 2016];83(3):436–43. Retrieved from: http://cardiovascres.oxfordjournals.org/content/83/3/436
- ↑ Hallett JW. Abdominal Aortic Aneurysms (AAA) [Internet]. Merck Manuals Professional Edition. Merck Sharp & Dohme Corp; 2016 [cited 2016Apr6]. Retrieved from: http://www.merckmanuals.com/professional/cardiovascular-disorders/diseases-of-the-aorta-and-its-branches/abdominal-aortic-aneurysms-(aaa)
- ↑ 17.0 17.1 Majeed K, Hamer A, White S, Pegg T, Wilkins G, Williams M, et al. Prevalence of abdominal aortic aneurysm in patients referred for transthoracic echocardiography. Internal Medicine Journal [serial on the Internet]. (2015, Jan), [cited March 24, 2016]; 45(1): 32-39 8p. Available from: CINAHL.
- ↑ Wikimedia https://commons.wikimedia.org/wiki/File:US_normal_abdominal_aorta.JPGfckLRAccessed April 5, 2016
- ↑ Wikimedia https://commons.wikimedia.org/wiki/File:RupturedAAAXray.png fckLRAccessed April 5, 2016
- ↑ https://commons.wikimedia.org/wiki/File:RupturedAAA.pngfckLRAccessed April 5, 2016
- ↑ WikipediafckLRhttps://en.wikipedia.org/wiki/Abdominal_aortic_aneurysmfckLRAccessed April 5, 2016
- ↑ 22.0 22.1 22.2 22.3 22.4 22.5 Hillegass EA. Essentials of Cardiopulmonary Physical Therapy. 3rd ed. St. Louis, MO: Elsevier/Saunders; 2011. 72, 397-398.
- ↑ Buck D, van Herwaarden J, Schermerhorn M, Moll F. Endovascular treatment of abdominal aortic aneurysms. Nat Rev Cardiol. 2014;11(5):250-250.
- ↑ 24.0 24.1 Schanzer A, Messina L. Two Decades of Endovascular Abdominal Aortic Aneurysm Repair: Enormous Progress With Serious Lessons Learned. Journal of the American Heart Association. 2012;1(3):e000075-e000075.
- ↑ Norwood M, Lloyd G, Bown M, Fishwick G, London N, Sayers R. Endovascular abdominal aortic aneurysm repair. Postgraduate Medical Journal. 2007;83(975):21-27.
- ↑ 26.0 26.1 MYERS J, MCELRATH M, JAFFE A, SMITH K, FONDA H, VU A et al. A Randomized Trial of Exercise Training in Abdominal Aortic Aneurysm Disease. Medicine & Science in Sports & Exercise. 2014;46(1):2-9.
- ↑ Wikimedia Commons https://commons.wikimedia.org/wiki/File:Gym_Cardio_Area.jpg Accessed April 9, 2016. Attribution: https://www.localfitness.com.au/
- ↑ Pouwels S, Willigendael E, van Sambeek M, Nienhuijs S, Cuypers P, Teijink J. Beneficial Effects of Pre-operative Exercise Therapy in Patients with an Abdominal Aortic Aneurysm: A Systematic Review. Journal of Vascular Surgery. 2015;61(1):284.
![[27]Cardiovascular fitness can help improve function in AAA patients](/images/thumb/5/52/Cardio.jpg/300px-Cardio.jpg)






