Angiotensin Inhibitors and Blockers for Treating Hypertension
Introduction[edit | edit source]
High blood pressure is a common condition in people. Long term high blood pressure may eventually cause health problems eg stroke, congestive heart failure. ACE inhibitors and angiotensin receptor blockers are a group of pharmaceuticals that can treat high blood pressure. These medications have similar mechanisms.[1]
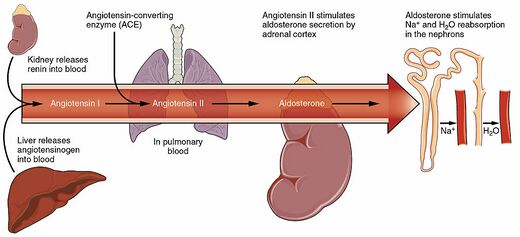
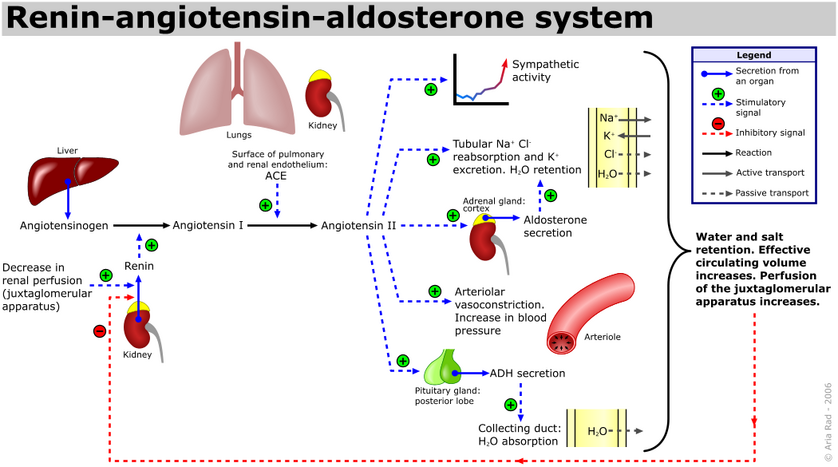
These drugs act by inhibiting the renin-angiotensin-aldosterone system (RAAS) and include angiotensin-converting enzyme (ACE) inhibitors and angiotensin-receptor blockers (ARBs).[2]
Watch this 2 minute video on ACE inhibitors.
History[edit | edit source]
Drugs that inhibit the renin-angiotensin system, such as angiotensin converting enzyme (ACE) inhibitors and angiotensin receptor antagonists, have proven value for the treatment of hypertension, heart failure and renal disease.
ACE inhibitors were invented first then ARBs were invented to prevent the side effects of ACE inhibitors. They reduce the rates of death, myocardial infarction and stroke in a broad range of patients at high risk, but do not control the blood pressure in all cases.
This led to research into inhibiting the renin-angiotensin system at its first step (the production of angiotensin) and a new type of medicine the direct renin inhibitors (marketed since 2009 to treat hypertension). Renin inhibitors represent an alternative strategy for inhibiting the renin-angiotensin system. They have a mechanism of action different from that of ACE inhibitors and angiotensin receptor antagonists. Whether renin inhibitors offer therapeutic benefits beyond those provided by ACE inhibitor and angiotensin receptor antagonist therapies will require their direct comparison in clinical outcome studies[4][5][6].
ACE inhibitors[edit | edit source]
ACE inhibitors block the production of angiotensin II, a substance that narrows blood vessels and releases hormones such as aldosterone and norepinephrine, by inhibiting an enzyme called angiotensin converting enzyme. Angiotensin II, aldosterone, and norepinephrine all increase blood pressure and urine production by the kidneys. If levels of these three substances decrease in the body, this allows blood vessels to relax and dilate (widen), reducing both blood and kidney pressure. ACE inhibitors also increase the production of bradykinin, another substance that makes blood vessels dilate[7].
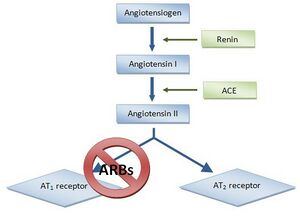
A common criticism of ACE-Inhibitors is that they fail to suppress angiotensin II alternative enzymatic pathways that counteract therapeutic effects.[8] As a result, Angiotensin II receptor blockers were introduced. Angiotensin II receptor blockers (ARB’s) are an angiotensin II antagonist at the type 1 receptor (AT1). ARB’s have a very high affinity for AT1 and these receptors are highly concentrated in tissues such as smooth muscle cells, kidneys, and the heart.[9]
Side effects of ACE inhibitors may include:
- Dry cough
- Increased potassium levels in the blood (hyperkalemia)
- Fatigue
- Hypotension
- Headaches
- Loss of taste[10]
ARBs[edit | edit source]
ARBs work by blocking the action of a natural chemical called angiotensin II. Angiotensin II is a potent vasoconstrictor. This narrowing can cause high blood pressure and poor blood flow through the kidneys. ARBs prevent angiotensin II from binding to angiotensin II receptors located in muscles surrounding blood vessels, thereby allowing those blood vessels to dilate, reducing blood pressure and improving symptoms of heart failure and progression of kidney disease due to diabetes. Angiotensin II also has effects on: noradrenaline (contributes to vasoconstriction and increases heart rate); aldosterone secretion from the adrenal cortex; and sodium re-absorption and water retention by the kidneys.[11]
Possible side effects of angiotensin II blockers can include:
- Dizziness
- Hyperkalemia
- Swelling of the skin due to a buildup of fluid (angioedema)[12]
Pharmacokinetics[edit | edit source]
ACE-Inhibitors
- Halve-lives range from 2.2 to 15 hours.
- Bind to tissues and plasma protein, which means that plasma concentrations are longer lasting and have a longer elimination phase.[9]
- Primarily excreted via the kidneys.
- Some ACE-Inhibitors are prodrugs, which means the compound is inactive until metabolized in the liver into the active substance allowing for increased absorption from the GI tract and prevention of the first-pass effect.
- Certain ACE-Inhibitors, as well as ARB’s, have been shown to be cardioprotective and cardioreparative via prevention and reversal of cardiac fibrosis.[13]
ARB’s
- Half-lives depends on specific drug
- Have a high absorption rate with oral administration, but their metabolism varies, some are active metabolites, and some are prodrugs.[9]
- Newer ARB’s are more potent with a slow dissociation, therefore they have longer half-lives and only require one daily dose as opposed to older ARB’s that require two doses to maintain BP levels.[8][14]
ACE-Inhibitors and ARB’s are not recommended for patients who are pregnant, or have a history of angioneurotic edema, hyperkalemia, or bilateral renal artery stenosis.[8]
See Also Pharmacological Management of Hypertension
References:[edit | edit source]
- ↑ Difference between Difference Between Ace Inhibitors and Angiotensin Receptor Blockers Available: https://www.differencebetween.com/difference-between-ace-inhibitors-and-angiotensin-receptor-blockers/ (accessed 29.3.2022)
- ↑ Amboss RAAS inhibitors Available from: https://www.amboss.com/us/knowledge/Renin-angiotensin-aldosterone_system_inhibitors (last accessed 20.6.2019)
- ↑ British Heart Foundation How do ACE inhibitors work? Available from: https://www.youtube.com/watch?v=xIlaQuRaZmk (last accessed 17.6.2019)
- ↑ NPS medwise Renin Inhibitors Available:https://www.nps.org.au/australian-prescriber/articles/renin-inhibitors-mechanisms-of-action (accessed 29.3.2022)
- ↑ Futura sciences Direct renin inhibitor Available: http://www.futura-sciences.us/dico/d/medical-science-direct-renin-inhibitor-50006488/ (accessed 29.3.2022)
- ↑ Australian Prescriber Renin inhibitors - mechanisms of action Available from: https://www.nps.org.au/australian-prescriber/articles/renin-inhibitors-mechanisms-of-action#conclusion (last accessed 20.6.2019)
- ↑ Drugs.com Angiotensin Converting Enzyme Inhibitors Available: https://www.drugs.com/drug-class/angiotensin-converting-enzyme-inhibitors.html (accessed 29.3.2022)
- ↑ 8.0 8.1 8.2 Laurent, S. Antihypertensive drugs. Pharmacological Research. October, 2017;124; 116-125. doi:10.1016/j.phrs.2017.07.026. Accessed November 29, 2018.
- ↑ 9.0 9.1 9.2 Therapeutics. British and Irish Hypertension Society. Retrieved from https://bihsoc.org/resources/therapeutics/. Accessed November 29, 2018.
- ↑ mayo clinic ACE inhibitors Available: https://www.mayoclinic.org/diseases-conditions/high-blood-pressure/in-depth/ace-inhibitors/art-20047480(accessed 29.3.2022)
- ↑ Drugs.com ARBs Available: https://www.drugs.com/drug-class/angiotensin-ii-inhibitors.html (accessed 29.3.2022)
- ↑ Mayo Clinic ARBs Available: https://www.mayoclinic.org/diseases-conditions/high-blood-pressure/in-depth/angiotensin-ii-receptor-blockers/art-20045009(accessed 29.3.2022)
- ↑ Kirsten, R, Nelson, K, Kirsten, D, Heintz, B. Clinical pharmacokinetics of vasodilators. Part I. Clinical Pharmacokinetics. June, 1998;34(6), 457–482. Retrieved from https://courseinfo.wssu.edu/login?url=http://search.ebscohost.com/login.aspx?direct=true&db=mdc&AN=9646008&site=ehost-live. Accessed November 29, 2018.
- ↑ Barreras, A, Gurk-Turner, C. Angiotensin II receptor blockers. Proceedings Baylor University Medical Center. January, 2003; 16(1), 123–126. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1200815/. Accessed November 29, 2018.