Necrotizing Fasciitis (Flesh Eating Disease)
Original Editor - Sara Halliday & Ashley Walker from Bellarmine University's Pathophysiology of Complex Patient Problems project.
Top Contributors - Ashley Walker, Sara Halliday, Elaine Lonnemann, Leana Louw, Shaimaa Eldib, Admin, Kim Jackson, Wendy Walker, WikiSysop, Lucinda hampton, Nupur Smit Shah and 127.0.0.1
Introduction[edit | edit source]
Necrotizing fasciitis is a potentially life-threatening bacterial infection that consists of rapidly progressing necrosis of fascia and subcutaneous tissues.[1][2][3] At first the tissues overlying are not affected, a possible cause of late diagnosis and surgical intervention.[4] The most rapidly progressing type of necrotizing fasciitis is Group A streptococcal infection, also known as flesh-eating bacteria. Necrotizing fasciits can also involve microbial infections with a singular bacteria (monomicrobial) or a combination of bacteria (polymicrobial).
Necrotizing fasciitis can occur due to several reasons (traumatic and non-traumatic) and in a variety of patient populations. Some conditions have been found to predispose patients to the risk of infection. Most of these conditions cause immunosuppression and include diabetes, malignancy, drug abuse, and chronic renal disease.[5]
The progression of the infection begins with the introduction of bacteria to the site and typically a result of trauma to the skin, however, trauma is not a necessary component. Once the infection is seeded locally, the bacteria spreads through deep fascial planes causing widespread tissue damage and infection. The spread of bacteria can cause ischemia to the area due to thrombosis occurring in blood vessels which can eventually result in gangrene.[6]
Epidemiology[edit | edit source]
Necrotizing fasciitis in some regions of the world is as common as one in every 100,000 people, compared to about 0.4 in every 100,000 people per year in the United States.[4] Necrotizing fasciitis is considered a rare condition, however, the mortality rate remains high. Evidence has estimated the mortality rate to be at 20-40%, increasing with the delay in initial diagnosis.[2][7]
Etiology[edit | edit source]
Necrotizing fasciitis is caused as a direct sequela of bacterial infection that enters the body through a break in the skin (in roughly 80% of all cases).The most common bacteria associated with necrotiing fasciitis after minor trauma is Group A Streptococcus. When necrotizing fasciitis develops after surgery, it is likely a mix of bacteria. Upon entering the body, it rapidly reproduces and emits toxins/enzymes that damage fascia and soft tissue. The bacteria will shield itself from the body's immune system which speeds up the proliferation along tissue planes.[8]
Characteristics/Clinical Presentation[edit | edit source]
The patient may initially present with:
- increasing pain,
- tenderness to palpation to the local area and periwound,
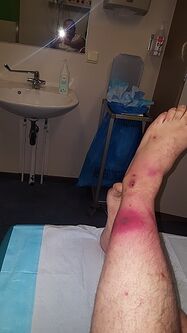
- purpura, erythema, swelling.[9]
- , crepitus, purulent drainage and usually a history of some type of trauma.[2]
- Complaints of flu-like symptoms (nausea, vomiting, fever, malaise etc.) are also common.
- Patients can present with symptoms of sepsis that include fever, tachycardia, altered mental state and diabetic ketoacidosis that typically accompanies trauma to the skin.[6]
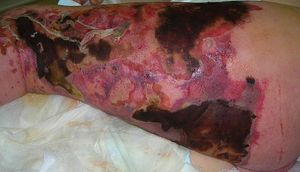
- Progressive symptoms include subcutaneous bleeding, gangrene and necrosis, which can lead to systemic inflammatory response syndrome (SIRS), septic shock and even death.[2]
Clinical presentation of necrotizing fasciitis varies and can typically be mistaken for other pathologies, such as cellulitis and superficial skin infections. Awareness of the presentation of necrotizing fasciitis clinically is critical due to the rapid progression of the disease. Evidence has shown that accurate diagnosis is difficult with only 15% to 34% of patients with necrotizing fasciitis having the appropriate admittion diagnosis.[6] Misdiagnosis often occurs due to the vague symptoms that may be present (i.e. erythema, fever, pain), or the lack thereof.
Types[edit | edit source]
Necrotizing fasciitis can be classified into 2 types:
- Type 1: is defined as necrotizing fasciitis caused by both aerobic and anaerobic bacteria,
- Tpe 2 :is caused by group A betahaemolytic Streptococcus and Staphylococcus aureus.[2]
Systemic Involvement[edit | edit source]
There are multiple systems that are compromised as the result of necrotizing fasciitis. If untreated, necrotizing fasciitis can lead to multi-system organ failure. Specific systems involved include:[10]
- Integumentary system: Tissue decay spawned from the infection
- Cardiovascular system: Implicated from the hypotension that occurs as a sign of necrotizing fasciitis. In addition, heart failure can result from advancement of the condition.
- Pulmonary system: Can be compromised from eventual respiratory failure that can occur in later stages
- Gastrointestinal system: Common sign of nausea and vomitting
- Genitourinary system: Can lead to renal failure
- Immunologic system: Comon sign of fever
Risk Factors:[edit | edit source]
- Diabetes (accosiated with 70.3% of patients in a 2003 study)[11]
- Chronic disease
- Immunosuppresion
- Intravenous drug use
- Peripheral vascular disease
- Renal failure
- Underlying malignancy[6]
Diagnostic Procedures:[edit | edit source]
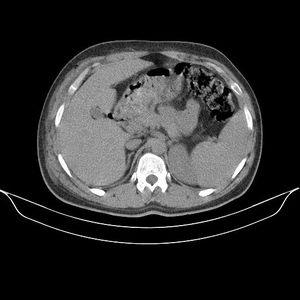
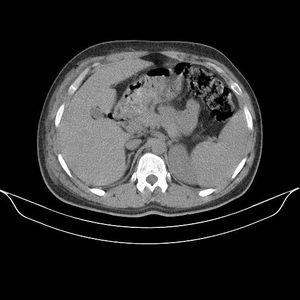
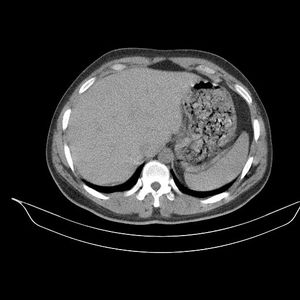
- CT scans: Deep fascial thickening, enhancement, fluid and gas in the soft tissue planes in and around superficial fascia are indicators of necrotizing faciitis.
- Ultrasound: Thickening and distortion of deep fascia, and fluid collections along deep fascia are denoted
- MRI: Deep fascial thickening, deep fascial fluid collections, and hyperintense T2W signal within muscles
- Controversial topic as some authors state that fascial enhancement is an indicator, while other authors state that lack of fascial enhancement is a positive finding.
- Laboratory tests: (same as ones used for severe soft tissue infections)
- Full blood count
- Electrolytes
- Erythrocyte sedimentation rate
- C-reactive protein
- Frozen section biopsy: Limited due to high costs involved[2]
A numerical score sheet, called the laboratory risk indicator for necrotizing fasciitis (LRINEC), was devised from lab parameters as a possible indicating tool for detection of necrotizing fasciitis. Score of ≥6 has a positive predictive value of 92% and a negative predictive value of 96%.[11]
Scoring of the LRINEC of less than 5 is indicative of a low risk of <50%; a score of 6-7 of an intermediate risk and a score of 8 and more indicates an high risk of more than 75%. The scoring is based on the following criteria:
| Parameter | Range | Score |
|---|---|---|
| Hb (g/dl) | >13.5
11.0 - 13.5 <11 |
0
1 2 |
| White cells (10^9/L) | <15
15 - 25 >25 |
0
1 2 |
| Sodium (mmlo/L) | <135 | 2 |
| Creatinine (μmol/L) | >141 | 2 |
| Glucose | >10 | 1 |
| C-reactive protein | >150 | 4 |
In addition, blood and pus cultures could be examined and surgical exploration may be indicated to confirm the diagnosis of necrotizing faciitis.[12] . However,the LRINEC scoring system lacked diagnostic sensitivity. Elevated serum lactate was supported as both a diagnostic and prognostic indicator[13]
Differential Diagnosis :[edit | edit source]
- Cellulitis[14]
- Superficial skin infections
- Acute epididymitis
- Gas gangrene
- Hernias
- Orchitis
- Testicular torsion
- Toxic shock syndrome
Medical Management:[edit | edit source]
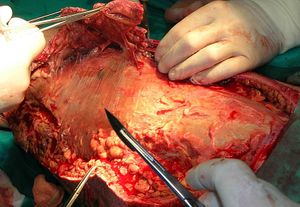
An aggressive form of treatment for necrotizing fasciitis is necessary due to the rapid progression of the infection once contracted. The typical management includes intravenous broad-spectrum antibiotics and early surgical debridement in order to remove the necrotic tissue.[6] Until surgical debridement has occurred, tissue hypoxia limits the efficacy of intravenous antibiotics.[6] A high dose antibiotic regimen is critical in the treatment of the infection in order to limit the amount of tissue damage that may occur. The inflammation that is associated with the infection causes intense pain and discomfort that is unrelieved by analgesic medications.[14] These two clinical findings present in necrotizing fasciitis assist to differentiate it from cellulitis. Early recognition is vital for successful treatment. If detected during the early stages, surgical debridement can be minor and more localized to the infected epidermis, subcutaneous and adipose tissue.[15]
Wound care is of utmost importance following surgical debridement[16]. Depending on the extent of the pathology, extensive debridement might be needed, resulting in big operative wounds. This holds a subsequent risk for further wound infections, and optimal wound care is of utmost importance. Vacuum dressings or negative pressure wound therapy might be used to facilitate the process of wound healing, by enhancing blood perfusion and promote granulation tissue, especially in the depth of the wound. This removes exudate, which decreases the inhibitory mediators and matrix metalloproteinases that interfere with the healing process.[17]
In the later stages of necrotizing fasciitis, systemic shock can occur resulting in failure of many organ systems. Respiratory failure, heart failure, low blood pressure and renal failure may occur in this stage. Limb amputation is necessary once the infection begins to spread to other organ systems.
Hyperbaric oxygen therapy has been proposed as an adjunct therapy for the treatment of necrotizing fasciitis. However, evidence has found conflicting results on the efficacy of this treatment. The main advantages of hyperbaric oxygen therapy in addition to standard regimes are tissue preservation and decreased mortality.[7] In another study by Golger et al, it was found that the addition of hyperbairc oxygen therapy showed no improvement in mortality rate.[18] A need exists for higher levels of research to be conducted for more conclusive results.
Physiotherapy Management:[edit | edit source]
Physiotherapy management will primarily occur after surgical debridement, and mainly focus on maintenance, prevention and management of secondary complications. Physiotherapy does not play a role in the curative management of this pathology. Patients with necrotizing fasciitis have an increased risk for the loss of endurance and strength.
- Prevention:
- Prophylactic chest physiotherapy to prevent atelectasis, as these patients often spend the most of their time in bed
- Loss of muscle power and range of motion: By incorporating exercise program
- Loss of function: Mobility program
- Pressure sores: Positioning and mobilization
- Maintenance: Of chest, function and range of motion
- Management of secondary complications:
- Pneumonia/chest infections/respiratory failure with the incorporation of chest physiotherapy
- Effects of immobility - Graded strengthening exercise program; transfer training; gait re-education; improve independence; address functional limitations
- Pressure sores: Positioning and mobilization
- Amputation rehabilitation[19][20]
References[edit | edit source]
- ↑ Shupak A, Oren S, Goldenberg I, Barzilai A, Moskuna R, Bursztein S. Necrotizing fasciitis: an indication for hyperbaric oxygenation therapy? Surgery 1995;118(5):873-8.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Bechar J, Sepehripour S, Hardwicke J, Filobbos G. Laboratory risk indicator for necrotising fasciitis (LRINEC) score for the assessment of early necrotising fasciitis: a systematic review of the literature. The Annals of The Royal College of Surgeons of England 2017 May 27;99(5):341-6.
- ↑ Schroder̈ A, Gerin A, Firth GB, Hoffmann KS, Grieve A, von Sochaczewski CO. A systematic review of necrotising fasciitis in children from its first description in 1930 to 2018. BMC infectious diseases. 2019 Dec;19(1):317.
- ↑ 4.0 4.1 Wallace HA, Perera TB. Necrotizing fasciitis. InStatPearls [Internet] 2021 Jul 27. StatPearls Publishing.Available:https://www.ncbi.nlm.nih.gov/books/NBK430756/ (accessed 2.2.2023)
- ↑ Hsiao C-T, Weng H-H, Yuan Y-D, Chen C-T, Chen I-C. Predictors of mortality in patients with necrotizing fasciitis. American Journal of Emergency Medicine 2008; 26: 170–75.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 Puvanendran R, Huey JC, Pasupathy S. Necrotizing fasciitis. Canadian family physician 2009;55(10):981-7.
- ↑ 7.0 7.1 Jallali N, Withey S, Butler PE. Hyperbaric oxygen as adjuvant therapy in the management of necrotizing fasciitis. The American Journal of Surgery 2005;189(4):462-6.
- ↑ The National Necrotizing Fasciitis Foundation. What is NF? Available from: http://www.nnff.com/what-is-nf.html (accessed 1 April 2012).
- ↑ Kiat HJ, Natalie YH, Fatimah L. Necrotizing fasciitis: How reliable are the cutaneous signs?. Journal of emergencies, trauma, and shock. 2017 Oct;10(4):205.
- ↑ Goodman C, Snyder T. Differential Diagnosis for Physical Therapists Screening for Referral. 4th ed. St. Louis: Saunders Elsevier, 2007.
- ↑ 11.0 11.1 Wong CH, Chang HC, Pasupathy S, Khin LW, Tan JL, Low CO. Necrotizing fasciitis: clinical presentation, microbiology, and determinants of mortality. J Bone Joint Surgery Am 2003;85-A(8):1454-60.
- ↑ Fazeli M, Keramati M. Necrotizing fascitis: an epidemiologic study of 102 cases. Indian Journal Of Surgery 2007;69:136-139.
- ↑ Hodgins N, Damkat-Thomas L, Shamsian N, Yew P, Lewis H, Khan K. Analysis of the increasing prevalence of necrotising fasciitis referrals to a regional plastic surgery unit: a retrospective case series. Journal of Plastic, Reconstructive & Aesthetic Surgery. 2015 Mar 1;68(3):304-11.
- ↑ 14.0 14.1 Bisno AL, Cockerill FR, Bermudez CT. The Initial Outpatient-Physician Encounter in Group A Streptococcal Necrotizing Fasciitis. Clinical Infectious Diseases 2000 Aug; 31:607–8.
- ↑ El-khani U, Nehme J, Darwish A, Jamnadas-Khoda B, Scerri G, Heppell S, Bennett N. Multifocal necrotising fasciitis: an overlooked entity?. Journal of Plastic, Reconstructive & Aesthetic Surgery. 2012 Apr 1;65(4):501-12.
- ↑ Corona PS, Erimeiku F, Reverté-Vinaixa MM, Soldado F, Amat C, Carrera L. Necrotising fasciitis of the extremities: implementation of new management technologies. Injury. 2016 Sep 1;47:S66-71.
- ↑ Baharestani M. Negative pressure wound therapy in the adjunctive management of necrotizing fascitis: examining clinical outcomes. Ostomy Wound Management 2008;54:44-50.
- ↑ Golger A, Ching S, Goldsmith CH, Pennie RA, Bain JR. Mortality in patients with necrotizing fasciitis. Plast Reconstr Surg 2007; 119(6): 1803-7.
- ↑ Duffin M, Farist K, Novak M. The impact of physical therapy on necrotizing fasciitis: A case report. Georgia Regent University. 2014.
- ↑ Vaughn A. Physical therapy intervention for in acute care for an adolescent with extensive muscle destruction follwoing necrotizing fasciitis: A case report [dissertation]. The Faculty of the Marieb College of Health and Human Services Florida Gulf Coast University. 2017.