Rheumatoid Arthritis
Original Editors - Florence Brachotte Amanda Fetz & Katie Robertson from Bellarmine University's Pathophysiology of Complex Patient Problems project
Top Contributors - Amanda Fetz, Riley Sturzebecher, Kim Jackson, Lucinda hampton, Amanda Ager, Admin, Aminat Abolade, Elaine Lonnemann, Wendy Walker, WikiSysop, Simisola Ajeyalemi, Shaimaa Eldib, Amrita Patro, 127.0.0.1 and Katie Robertson Amanda Fetz,Riley Sturzebecher,Elaine Lonnemann,Rachael Lowe
Introduction[edit | edit source]
Rheumatoid arthritis (RA) is a systemic autoimmune disease characterized by inflammatory arthritis and extra-articular involvement. RA with symptom duration of fewer than six months is defined as early, and when the symptoms have been present for more than months, it is defined as established.
There is no laboratory test that is pathognomonic for rheumatoid arthritis. The treatment of patients with rheumatoid arthritis requires both pharmacological and non-pharmacological agents. Today, the standard of care is early treatment with disease modifying anti-rheumatic drugs[1]
Etiology and Pathology[edit | edit source]
Etiology is unknown (probably multifactorial).
- It is generally considered that a genetic predisposition (e.g. HLA-DR B1 which is the most common allele of HLA-DR4 involved in rheumatoid arthritis) and an environmental trigger (e.g. Epstein-Barr virus postulated as a possible antigen, but not proven) lead to an autoimmune response that is directed against synovial structures and other organs.
Activation and accumulation of CD4 T cells in the synovium start a cascade of inflammatory responses which result in:
- activation of the macrophages and synovial cells and production of cytokines (eg L4 and TNF,) which in turn cause proliferation of the synovial cells and increase the production of destructive enzymes (eg elastase and collagenase) by macrophages
- activating B cell lymphocytes to produce various antibodies (including rheumatoid factor) which makes immune complexes that deposit in different tissues and contribute to further injury
- directly activate endothelial cells via increased production of VCAM1, which increases the adhesion and accumulation of inflammatory cells
- producing RANKL which in turn activate osteoclasts causing subchondral bone destruction
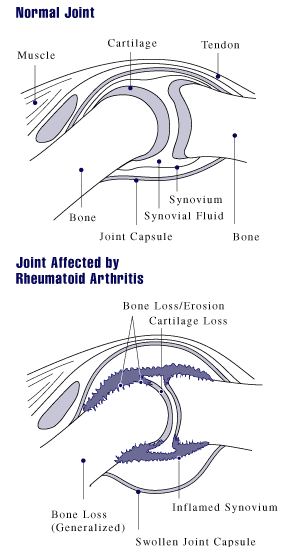
The inflammatory response leads to Pannus formation.
Pannus:
- Is an oedematous thickened hyperplastic synovium infiltrated by lymphocytes T and B, plasmocytes, macrophages, and osteoclasts.
- It will gradually erode bare areas initially, followed by the articular cartilage.
- Goes on to causes fibrous ankylosis which eventually ossifies[2]
Epidemiology[edit | edit source]
The overall prevalence is 0.5-1% and the disease is 2-3 times more common in women.
Onset is generally in adulthood, peaking in the 4th and 5th decades. The paediatric condition, Juvenile Idiopathic Arthritis (JIA), is discussed separately, see link.[2]
Risk Factors[edit | edit source]
Include:
- Increased prevalence of RA within families resulting from the interaction between patients genotype and environment.
- Increases risk: Female sex; occupational dust (silica); air pollution; high sodium red and iron consumption; low vitamin D intake and levels; Smoking (seropositive RA); Obesity; Low socioeconomic status [2]
- Deceased risk: fish and omega 3 fatty acid consumption; moderate alcohol intake; healthy diet; oral contraceptive/HRT; statin use.[3]
Characteristics/Clinical Presentation[edit | edit source]
In rheumatoid arthritis, joint complaints are in the foreground. The most common clinical presentation of RA is
- Polyarthritis of small joints of hands: proximal interphalangeal (PIP), metacarpophalangeal (MCP) joints and wrist. Some patients may present with monoarticular joint involvement.
- Commonly joint involvement occurs insidiously over a period of months, however, in some cases, joint involvement may occur over weeks or overnight.
- Other commonly affected joints include wrist, elbows, shoulders, hips, knees, ankles and metatarsophalangeal (MTP) joints.
- Stiffness in the joints in the morning may last up to several hours, usually greater than an hour. The patient may have a "trigger finger" due to flexor tenosynovitis.
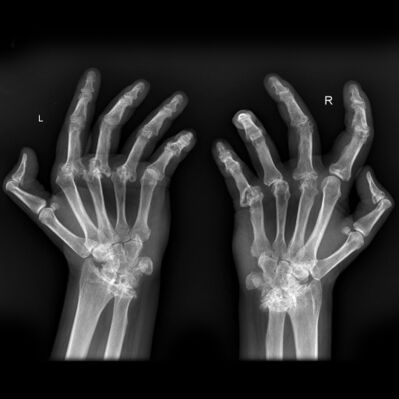
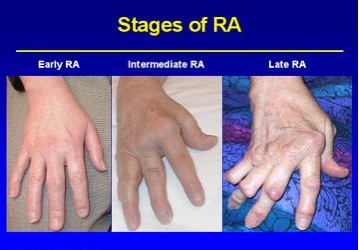
Image 4: Advanced features of rheumatoid arthritis, with erosive subluxation most marked of the MCP joints with ulnar deviation. Prominent degenerative change is also seen at the ulnar-carpal articulation. Note also osteopenia particularly of the MCP regions. On examination,
- May be swelling, stiffness, deformity, and tenderness of the PIP, MCP wrist, knee joints, referred to as synovitis, and there may be a decreased range of motion.
- Deformity, pain, weakness and restricted mobility resulting in loss of function.[4]
- Rheumatoid nodules may be present in 20% of patients with rheumatoid arthritis; these occur over extensor surfaces at elbows, heels, and toes.
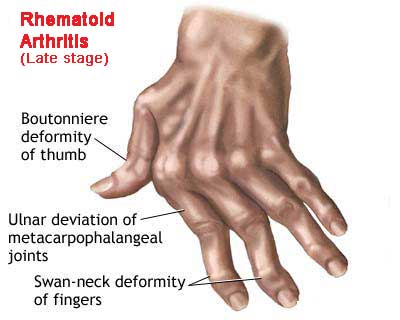
- Late in the course of the disease patient may present with "boutonniere (flexion at PIP and extension at DIP), swan neck (flexion at DIP and extension at PIP) deformities, subluxation of MCP joints and ulnar deviation.
- Other features may include the presence of carpal tunnel syndrome, tenosynovitis and finger deformities.
- Examine the joints on swelling, pain due to palpation, pain due to movement, decreased range of motion, deformation and instability.
- Hallmark symptoms such as symmetrical joint swelling and tenderness, morning stiffness, positive rheumatoid factor (RF), elevated acute phase reactants, and radiographic evidence of erosive bone loss.
- Significant predictors of functional decline among persons with RA are slow gait and a weak grip. [5][6]
Rheumatoid arthritis can affect almost every organ in the body
- The three most important complaints are pain, morning stiffness and fatigue.
- Muscular strength, muscular endurance and aerobic endurance are typically reduced in patients with rheumatoid arthritis in comparison with healthy patients.
- In 80-90% of the patients with rheumatoid arthritis the cervical spine is involved, which can lead to instability, caused by the ligamentous laxity (between the first and second cervical vertebrae most commonly) This instability can lead to pain and neurological symptoms, eg headache and tingling in the fingers. [5]
- Individuals with RA are 8 times more likely to have functional disability compared with adults in the general population from the same community.
Staging[edit | edit source]
Disease progression:
Stages
- No destructive changes on x-rays
- Presence of x-ray evidence of periarticular osteoporosis, subchondral bone destruction but no joint deformity
- X-ray evidence of cartilage and bone destruction in addition to joint deformity and periarticular osteoporosis.
- Presence of bony or fibrous ankylosis along with stage 3 features.[1]
Differential Diagnosis[edit | edit source]
- Lupus
- Chronic Lyme disease
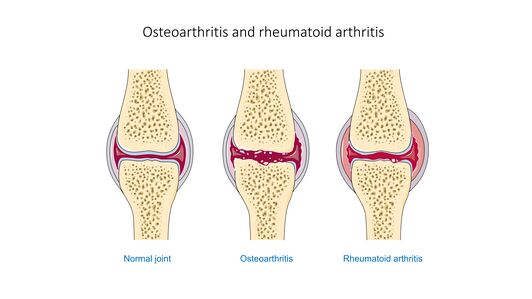
- Osteoarthritis
- Septic arthritis
- Psoriatic arthritis
- Sjogren syndrome
- Sarcoidosis[1]
Complications[edit | edit source]
RA has many effects on individuals including mortality, hospitalization, work disability, increase in medical cost/expenses, decreases of quality of life, and chronic pain. On average, the chronic RA patient has two or more comorbid conditions.[7] This is significant because of the comorbidities effects on quality of life, functional status, prognosis and outcome. Associated Complications include:[8]
- Infections
- Chronic anaemia
- Gastrointestinal cancers
- Pleural effusions
- Osteoporosis
- Heart disease
- Sicca syndrome
- Felty syndrome
- Lymphoma[1]
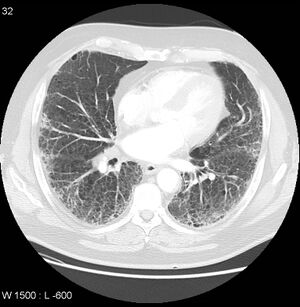
- Damage to the lung tissue (rheumatoid lung) See image 6: CT demonstrates extensive pulmonary fibrosis in the mid and lower zones with co-existing severe centrilobular emphysema in the upper zones. The fibrosis is attributable to the patient's known rheumatoid arthritis, and the emphysema to a long history of smoking.
- Side effects from treatment and medication.
- General deconditioning
- Neurological complications
- Ocular complications
Diagnostic Procedures[edit | edit source]
- Lab evaluation of patients with rheumatoid arthritis consists of obtaining
- Rheumatoid factor (antibody against the Fc portion of IgG). About 45% to 75% of patients with RA test positive for rheumatoid factor. However, the presence of rheumatoid factor is not diagnostic of rheumatoid arthritis. It may be present in connective tissue disease, chronic infections, and healthy individuals, mostly in low titers.
- Anti-citrullinated protein antibodies (ACPA) are found in about 50% of patients with early arthritis, which subsequently are diagnosed with RA.
- Acute-phase reactants, erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) may be elevated in the active phase of arthritis.
2. X-ray of both hands and feet are usually obtained for the presence of erosions, the pathognomonic feature of rheumatoid arthritis (plain radiograph does not show early changes of the disease).
3. Magnetic resonance imaging (MRI) and ultrasound of joints detect erosions earlier than an x-ray. MRI and US are more sensitive than clinical examination in identifying synovitis and joint effusion.[1]
Prognosis[edit | edit source]
Rheumatoid arthritis has no cure and is a progressive disease. All individuals have multiple exacerbations and remissions. Close to 50% of patients with the disease become disabled within 10 years.
- Besides the joint disease, the individuals can suffer from many extra joint-related problems which significantly alters the quality of life. The progression of disease does vary from individual to individual.
- Rheumatoid arthritis is also associated with cardiovascular risk factors, infection, respiratory disease and the development of malignancies. Patients with rheumatoid arthritis have a 2-3 times higher risk of death compared to the general population.[1]
Treatment[edit | edit source]
Treatment of rheumatoid arthritis is aimed at improving the symptoms and slowing disease progression.
Because the disorder affects many other organs, it is best managed with an inter-professional team. The key is patient education by nurses, pharmacists, and primary care providers. The nurse should inform the patient about the signs and symptoms of different organ systems and when to seek medical care. A physiotherapist should implement an exercise program to recover joint function. An occupational therapy consult can help the patient manage daily living activities. The pharmacist should educate the patient on the types of drugs used to treat rheumatoid arthritis and their potential side effects.
Medical Management[edit | edit source]
Medications used in treatment include:
- non-steroidal anti-inflammatory drugs (NSAIDs)
- disease-modifying anti-rheumatic drugs (DMARDs)
- conventional synthetic DMARDs (csDMARDs): e.g. methotrexate, leflunomide, prednisolone
- biological DMARDs (bDMARDs): e.g. TNF-α inhibitors (e.g. infliximab), tocilizumab, abatacept, rituxumab
- targeted synthetic DMARDs (tsDMARDs): e.g. tofacitinib
The disease carries a significant burden of disability. There is also a reduction in life expectancy, with excess mortality usually related to its non-articular manifestations.[2]
See link Pharmacological Management of Rheumatoid Arthritis
Nutritional Guidelines[edit | edit source]
Dietary interventions demonstrate substantial benefits in reducing disease symptoms such as pain, joint stiffness, swelling, tenderness and associated disability with disease progression (still an uncertainty about the therapeutic benefits of dietary manipulations for RA)[9]. Dietary modification include:
- Avoiding food that causes inflammation like processed food, high salt, oils, butter, sugar, and animal products.
- Supplements: Research suggests that there are vitamins and minerals which may have an effect on RA[9]eg. vitamin D, cod liver oil, and multivitamins. These may help eg reduce joint inflammation, improve bone health. It is recommended to consult your primary care physician.
Physical Therapy Management[edit | edit source]
Rheumatoid arthritis is a chronic disorder that has no cure. All the currently available treatments are geared towards improving the symptoms and offering a better quality of life.[1] Treatments that achieve pain relief and the slowdown of the activity of RA to prevent disability and increase functional capacity.[10][11]
See also Hand RA
The benefits of physical therapy interventions have been well documented.
- Physical therapists play an integral role in the nonpharmacologic management of RA.
- Physiotherapy help clients cope with chronic pain and disability through the design of programs that address flexibility, endurance, aerobic condition, range of motion (ROM), strength, bone integrity, coordination, balance and risk of falls.
- All current UK clinical guidelines for the management of RA recommend the use of physiotherapy (PT) and occupational therapy (OT) as an adjunct to drug treatment.
The four most common components of PT/OT for RA hands are
- Exercise therapy,
- Joint protection advice and provision of functional splinting and assistive devices
- Massage therapy, and
- Patient education.
The therapy goals in most cases are: [12]
- Improvement in disease management knowledge
- Pain control
- Improvement in activities of daily living
- Improvement in Joint stiffness (~ Range of motion)
- Prevent or control joint damage
- Improve strength
- Improve fatigue levels
- Improve the quality of life
- Improve aerobic condition
- Improve stability and coordination
Patient questionnaires, not joint counts, radiographic scores, or laboratory tests, provide the most significant predictors of severe 5-year outcomes in patients with RA, including functional status, work disability, costs, joint replacement surgery and premature death.
Treatment techniques[edit | edit source]
Cold/Hot Applications: cold for acute phase; heat for chronic phase and used before exercise. [13]
Transcutaneous electrical nerve stimulation (TENS) is used to relieve pain. [14]
Hydrotherapy-Balneotherapy: exercise with minimal load on the joints.[15]
Joint Protection see link
Massage Therapy: Massage and the manual trigger of an articular movement focused on the improvement of function, pain reduction, reduction of disease activity improve flexibility and welfare (dimension of depression, anxiety, mood and pain) [16]
Therapeutic Exercise[edit | edit source]
- Physical exercise helps to increase the physical capacity of the patient [10].
- Exercise improves general muscular endurance and strength without detrimental effects on disease activity or pain in RA
- Before beginning an exercise program perform a global evaluation of the situation: joint-inflammation local or systemic, state of the disease, age of the patient and grade of collaboration.[10]
- Exercise therapy is aimed at improving daily functioning and social participation by means of improvement of the strength, aerobic condition, range of motion, stabilisation and coordination.
Programs for Patients with RA
- Includes; ROM-exercises; aerobic exercise: stabilisation/coordination exercises.
- Start with a moderate-intensive exercise program [17][18]
- Progress to a high-intensive exercise program if possible aimed at improving aerobic capacity, strength and endurance.
- The duration and intensity of the exercises should be based on the individual patient and their assessment[10].
Precautions[10][edit | edit source]
- When the patient experiences an exacerbation and the joints are acutely inflamed then isometric exercises should be done
- Avoid stretching in acute cases.
- Revise the exercise program if pain persists 2 hours after the activity or there is an increase in joint swelling
- Patients with active RA in their knees should avoid climbing stairs or weight lifting as it could lead to intra-articular pressure in the knee joint
- Avoid excessive stress over the tendons with stretches and avoid ballistic movements
Exercises examples[edit | edit source]
- In acute phase: isometric/static exercises -> be held for 6 seconds and repeated 5–10 times each day ; load = 40% 1RM. Chronic phase -> minimum 4 repetitions for each joint in 2 to 3 days These exercises increase the mobility of the joint, but the concerned joint will not be loaded during this exercises.[19] Contractures can be held for 6seconds and repeated 5-10 times daily[10].
- Stretching: Avoid in acute cases.
- Strengthening
- Aerobic condition exercises: There are two types of exercises to improve the aerobic condition: Intensive exercises and moderate-intensive exercises. The intensive exercise therapy has a minimum duration of 20 minutes per session and this 3 times a week with an intensity of 65 to 90 percent of the maximal heart rate. The moderate-intensive exercise therapy has a minimum duration of 30 minutes per session and this 5 times a week with an intensity of 55 to 64 percent of the maximal heart rate. The aim of these exercises is to improve muscle endurance and aerobic capacity. eg: swimming, walking, cycling
- Stabilizing and coordinating exercises: The improvement of stabilization and coordination of a certain joint will be achieved by doing exercises that stimulate the sensorimotor system. For example, standing on a balance board. Important aspects during these exercises are motion control, balance and coordination.
- Conditioning exercises in people with chronic inactive RA: swimming walking, cycling (include adequate rest periods)[10].
- Routine daily activities: SARAH (Strengthening and stretching for rheumatoid arthritis of the hand) exercise program see below table: The SARAH trial tests an intervention against the usual hand care. The main aim of the exercise program is increased hand function, which is suggested to be mediated by increases in strength, dexterity and range-of-movement. The exercise program consists of the usual care plus a hand and wrist exercise program which includes seven mobility exercises and four strength exercises against resistance (i.e. therapy putty, theraband or hand exerciser balls).[4]
| Exercises | Frequency | Sets | Repetitions | Initial Hold | Initial Load | Progression | |
| Mobility | • MCP flexion • Tendon gliding • Finger radial walking • Wrist circumduction • Finger adduction • Hand-behind-head • Hand-behind-back |
Daily | 1 | x 5 | 5 seconds (where required) | Step 1: increase up to 10 repetitions Step 2: Increase up to 10 seconds hold | |
| Strength | • Eccentric wrist extension • Gross grip Finger adduction • Pinch grip |
Daily | 1 | X 8 (min. 8 repetitions, max. 12 repetitions) | Between 3 and 4 on modified 10 pt Borg Scale | Step 1: 2x 10 repetitions Step 2: 4-5 on Borg scale Step 3: 5-6 on Borg scale Step 4: 3x 10 repetitions |
- Use modified Borg scale to set the load (resistance) for the strength exercises based on self-perception of effort.
- The level of resistance is determined by the patients’ rating of perceived effort using the weaker hand for each strength exercise.
- Exercise therapy in patients with RA is used to improve daily functioning and social participation through improving muscle strength, aerobic endurance, joint mobility and stability and/or coordination.
- Preference is given to an active policy, especially where the physiotherapist has a supporting role.
- In individual cases, passive treatments, such as manual operations, can be part of the treatment.
5. Patient Education: information about their condition and the different therapies disposed to improve their quality of life. eg Patients are taught how to protect the joints during routine daily life; adjusting their movement-behaviour; behavioural change by your patient (a process with 3 phases: the motivation-phase, the initial-behavioural change phase and the phase where the intended behaviour is continued).
• Formulate achievable goals with the patient.
• Give proper instructions and be sure that the patient understands.
• Enough variation in the exercises is important to prevent boredom.
• The therapist has to involve the partner and other important people in the process because they have an important motivation-role. Also, the therapist himself has to motivate the patient.
• Keep in touch with the patient to be sure that the treatment was effective. [20]
Management of flare ups
People who are diagnosed with RA also may experience a phenomenon that is called an “flare up”. Usually happen after eg experiencing a secondary illness, being involved in a high-stress situation, overexerting oneself, What triggers flare ups is currently still unknown. Strategies that can help someone who is experiencing a flare up
- Balance is key, schedule plenty of down time to reduce the likelihood of affected joints from becoming flared up
- Educate family, staff at work, and other people who you interact with, they can help you during flare ups
- Have a backup plan, be prepared in case of a flare up and become familiar with warning signs of a flare up
- Practice relaxation and self calming strategies: Research suggests that regularly practicing these relaxation techniques can reduce stress and lead to a reduction in pain.
- Use modalities such as a cold pack or hot pack: Both of these have various effects on tissues which research suggests can be beneficial in reducing inflammation and pain during a flare up
- Lastly, corticosteroid injections can be used to reduce inflammation and reduce pain in a flared up joint
Outcome Measures[edit | edit source]
- Simplified disease activity (SDAI) index: tender joint count, swollen joint count, patient global assessment, physician global assessment and c reactive protein in mg/dl
- Clinical disease activity index (CDAI): tender joint count, swollen joint count, patient global assessment, physician global assessment
- DAS28-ESR (disease activity score): tender joint count, swollen joint count, patient global assessment, and erythrocyte sedimentation rate in mm
- DAS-Crp (disease activity score): tender joint count, swollen joint count, patient global assessment, and c reactive protein in mg/dl.[1]
- Rheumatoid Arthritis Disease Activity Index (RADAI-5): A self-reported outcome measure that consists of 5 questions in a Likert scale format that briefly surveys the patient regarding their views of their condition (both over the past 6 months and current status).
- DASH
- 36-Item Short Form
- Fatigue Severity Scale
Classification of Functional Status[edit | edit source]
The American College of Rheumatology classified functional status in Rheumatoid Arthritis as:
- Class I: Completely able to perform usual activities of daily living (self-care, vocational, and avocational)
- Class II: Able to perform usual self-care and vocational activities, but limited in avocational activities
- Class III: Able to perform usual self-care activities, but limited in vocational and avocational activities
- Class IV: Limited ability to perform usual self-care, vocational, and avocational activities
Resources[edit | edit source]
- Rheumatoid Arthritis: Help to understand Rheumatoid Arthritis
- Rheumatoid Arthritis: Frequently asked Questions
- Rheumatology Check List Visit
- The RA Symptom Tracker Sheet
- The Arthritis Organization
- American College of Rheumatology Patient Education
- Self-help (Aids for Arthritis)
- RA Treatments
- American College of Rheumatology
Clinical Bottom Line[edit | edit source]
The outcome of most patients with Rheumatoid arthritis is guarded.
- The disorder has frequent relapses and remissions, and at least 40% of patients will become disabled within ten years.
- Some patients have mild disease, others may have a severe disease that severely affects the quality of life.
- Worse outcomes are usually seen in patients with a high titer of autoantibodies, HLA-DRB1 genotypes, age younger than 30, multiple joint involvement, female gender, and extra-articular involvement.
- The drugs used to treat rheumatoid arthritis also have potent side effects which often are not well tolerated. As the disease progresses, many patients will develop adverse cardiac events leading to death.
- The overall mortality in patients with rheumatoid arthritis is three times higher than in the general population.
- Despite advances in care, mortality from infection, cancer, and ongoing vasculitis remains unchanged[1]
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Krati Chauhan; Jagmohan S. Jandu; Mohammed A. Al-Dhahir. Oct 2019 RA Available from:https://www.ncbi.nlm.nih.gov/books/NBK441999/ (last accessed 23.2.2020)
- ↑ 2.0 2.1 2.2 2.3 Radiopedia RA Available from: https://radiopaedia.org/articles/rheumatoid-arthritis(accessed 7.6.2021)
- ↑ Deane KD, Demoruelle MK, Kelmenson LB, Kuhn KA, Norris JM, Holers VM. Genetic and environmental risk factors for rheumatoid arthritis. Best practice & research Clinical rheumatology. 2017 Feb 1;31(1):3-18.
- ↑ 4.0 4.1 Adams J, Bridle C, Dosanjh S, Heine P, Lamb SE, Lord J, McConkey C, Nichols V, Toye F, Underwood MR, Williams MA. Strengthening and stretching for rheumatoid arthritis of the hand (SARAH): design of a randomised controlled trial of a hand and upper limb exercise intervention-ISRCTN89936343. BMC musculoskeletal disorders. 2012 Dec;13(1):1-0.
- ↑ 5.0 5.1 KNGF-richtlijn. Reumatoïde artritis. 2008
- ↑ Neuberger GB, Aaronson LS, Gajewski B, Embretson SE, Cagle PE, Loudon JK, Miller PA. Predictors of exercise and effects of exercise on symptoms, function, aerobic fitness, and disease outcomes of rheumatoid arthritis. Arthritis Care & Research. 2007 Aug 15;57(6):943-52.
- ↑ Pubmed. Comorbidities in rheumatoid arthritis. http://www.ncbi.nlm.nih.gov/pubmed/17870034 (accessed 12 February 2013).
- ↑ Gabriel SE. Cardiovascular morbidity and mortality in rheumatoid arthritis. The American journal of medicine. 2008 Oct 1;121(10):S9-14.
- ↑ 9.0 9.1 Khanna S, Jaiswal KS, Gupta B. Managing rheumatoid arthritis with dietary interventions. Frontiers in nutrition. 2017 Nov 8;4:52.
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 Kavuncu V, Evcik D. Physiotherapy in rheumatoid arthritis. Medscape General Medicine. 2004;6(2).
- ↑ Williams MA, Srikesavan C, Heine PJ, Bruce J, Brosseau L, Hoxey‐Thomas N, Lamb SE. Exercise for rheumatoid arthritis of the hand. Cochrane Database of Systematic Reviews. 2018(7).
- ↑ Bell MJ, Lineker SC, Wilkins AL, Goldsmith CH, Badley EM. A randomized controlled trial to evaluate the efficacy of community based physical therapy in the treatment of people with rheumatoid arthritis. The Journal of rheumatology. 1998 Feb 1;25(2):231-7.
- ↑ Bijlsma JW, Geusens PP, Kallenberg CG, Tak PP. Reumatologie en klinische immunologie.
- ↑ Pelland L, Brosseau L, Casimiro L, Welch V, Tugwell P, Wells GA. Electrical stimulation for the treatment of rheumatoid arthritis. Cochrane Database of Systematic Reviews. 2002(2).
- ↑ Verhagen AP, Bierma‐Zeinstra SM, Boers M, Cardoso JR, Lambeck J, de Bie R, de Vet HC. Balneotherapy for rheumatoid arthritis. Cochrane Database of Systematic Reviews. 2004(1).
- ↑ Brownfield A. Aromatherapy in arthritis: a study. Nursing Standard (through 2013). 1998 Oct 21;13(5):34.
- ↑ de Jong Z, Munneke M, Zwinderman AH, Kroon HM, Jansen A, Ronday KH, van Schaardenburg D, Dijkmans BA, Van den Ende CH, Breedveld FC, Vlieland TP. Is a long‐term high‐intensity exercise program effective and safe in patients with rheumatoid arthritis?: results of a randomized controlled trial. Arthritis & Rheumatism: Official Journal of the American College of Rheumatology. 2003 Sep;48(9):2415-24.
- ↑ Van Den Ende CH, TP VV, Munneke M, Hazes JM. Dynamic exercise therapy for rheumatoid arthritis. The Cochrane database of systematic reviews. 2000 Jan 1(2):CD000322-.
- ↑ Minor MA, Webel RR, Kay DR, Hewett JE, Anderson SK. Efficacy of physical conditioning exercise in patients with rheumatoid arthritis and osteoarthritis. Arthritis & Rheumatism: Official Journal of the American College of Rheumatology. 1989 Nov;32(11):1396-405.
- ↑ Brodin N, Eurenius E, Jensen I, Nisell R, Opava CH, PARA Study Group. Coaching patients with early rheumatoid arthritis to healthy physical activity: a multicenter, randomized, controlled study. Arthritis Care & Research. 2008 Mar 15;59(3):325-31.
- ↑ O’Sullivan and Schmitz. Physical Rehabilitation. 5th edition. Philadelphia, PA: F.A. Davis Company. 2007.