Wound Healing
Original Editor - Esraa Mohamed Abdullzaher
Top Contributors - Esraa Mohamed Abdullzaher, Naomi O'Reilly, Lucinda hampton, Kim Jackson, Olajumoke Ogunleye, Joao Costa, Claire Knott, Lauren Lopez, Aya Alhindi and Admin
Introduction[edit | edit source]
Skin is the largest organ in the body and covers the body's entire external surface.
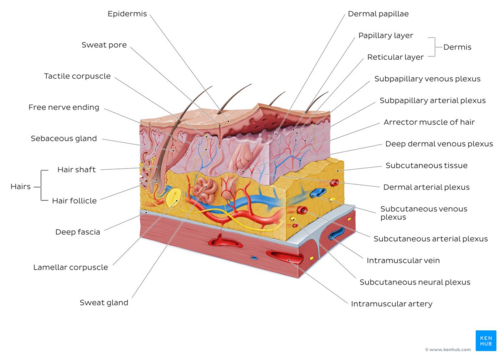
- Made up of three layers, the epidermis, dermis, and hypo-dermis.
- Skin's structure is made up of an intricate network that serves as the body’s initial barrier against pathogens, UV light, and chemicals, and mechanical injury, and regulates temperature and the amount of water released into the environment.
A skin wound results from the breakdown of the epidermal layer integrity[2].
- Wound healing mostly means healing of the skin.
- Begins immediately after an injury to the epidermal layer and might take years.
- Dynamic process including highly organized cellular, humoral, and molecular mechanisms.
- Has 3 overlapping phases which are inflammation, proliferation, and remodelling. Any disruption leads to abnormal wound healing[3].
See also Soft Tissue Healing
Types of Wounds[edit | edit source]
The definition of a wound in general is damage to the integrity of biological tissue, including skin, mucous membranes, and organ tissues.Wounds can be separated into open or closed wounds:
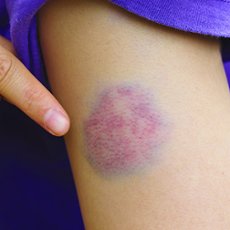
- Closed Wound: The surface of the skin is intact, but the underlying tissues may be damaged. e.g. contusions, haematomas, or Stage 1 Pressure Ulcers.
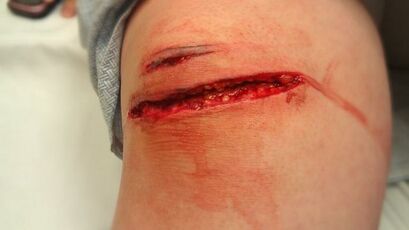
- Open Wounds: the skin is split or cracked and the underlying tissues are exposed to the outside environment.
Also it can be classified according to the cleanliness and condition of wounds into four classes of wound status:[4]
Class 1
wounds are considered to be:
- clean
- uninfected
- no inflammation is present
- primarily closed
- these wounds do not enter respiratory, alimentary, genital, or urinary tracts.
Class 2
wounds are considered to be:
- clean-contaminated
- lack unusual contamination
- Class 2 wounds enter the respiratory, alimentary, genital, or urinary tracts under controlled conditions.
Class 3
wounds are considered to be:
- contaminated
- These are fresh, open wounds caused by an insult to sterile techniques or by gastrointestinal tract leakage into the wound.
- incisions that result in acute or lack of purulent inflammation are classified as class 3 wounds.
Class 4
wounds are considered to be:
- dirty-infected
- result from improperly cared for traumatic wounds.
- demonstrate devitalized tissue
Types of Wound Healing[edit | edit source]
Wound healing is classified as primary, secondary, and tertiary wound healing.
Primary Healing or primary intention
- Uncomplicated healing of a non-infected, well-approximated wound is defined as primary healing. e.g. Surgical wounds.
Secondary Healing or secondary intention
- If the wound healing course in this wound is disrupted by infection, dehiscence, hypoxia or immune dysfunction, the secondary healing stage begins.
- During secondary healing, granulation tissue formation and epithelization over this new tissue take place. These types of wounds are more susceptible to infections and poor healing.
Tertiary healing or third intention
- it is delayed primary wound healing after 4–6 days.
- This occurs when the process of secondary intention is intentionally interrupted and the wound is mechanically closed.
- This usually occurs after granulation tissue has formed.
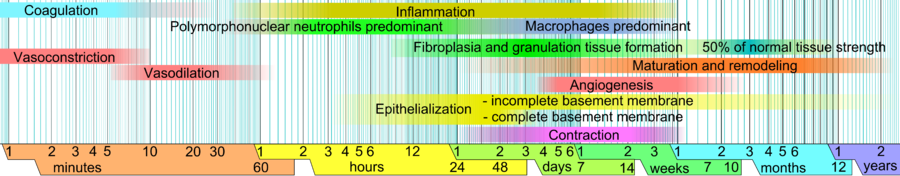
Wound Healing Stages in Adults[edit | edit source]
In adults, optimal wound healing should involve four continuous and overlapping phases: Haemostasis, inflammation, proliferation, and remodelling .[5]
Hemostasis Phase[edit | edit source]
The process of the wound being closed by clotting.
- Happens very quickly.
- Starts when blood leaks out of the body, then blood vessels constrict to restrict the blood flow.
- The platelets aggregate and adhere to the sub-endothelium surface within seconds of the rupture of a blood vessel's epithelial wall.
- After that, the first fibrin strands begin to adhere in about sixty seconds.
- As the fibrin mesh begins, the blood is transformed from liquid to gel through pro-coagulants and the release of prothrombin.
- The formation of a thrombus or clot keeps the platelets and blood cells trapped in the wound area.
- The thrombus is generally important in the stages of wound healing but becomes a problem if it detaches from the vessel wall and goes through the circulatory system, possibly causing a stroke, pulmonary embolism or heart attack[6][3].
Inflammatory Phase[edit | edit source]
- Begins right after the injury when the injured blood vessels leak transudate (made of water, salt, and protein) causing localized swelling.
- Inflammation both controls bleeding and prevents infection.
- The fluid engorgement allows healing and repair cells to move to the site of the wound.
- During the inflammatory phase, damaged cells, pathogens, and bacteria are removed from the wound area.
- The white blood cells, growth factors, nutrients and enzymes create the swelling, heat, pain and redness commonly seen during this stage of wound healing.
- Inflammation is a natural part of the wound healing process and is only problematic if prolonged or excessive.
Proliferative Phase[edit | edit source]
When the wound is rebuilt with new tissue made up of collagen and extracellular matrix
- The wound contracts as new tissues are built.
- A new network of blood vessels must be constructed so that the granulation tissue can be healthy and receive sufficient oxygen and nutrients.
- Myofibroblasts cause the wound to contract by gripping the wound edges and pulling them together using a mechanism similar to that of smooth muscle cells.
- In healthy stages of wound healing, granulation tissue is pink or red and uneven in texture. Healthy granulation tissue does not bleed easily.
- Dark granulation tissue can be a sign of infection, ischemia, or poor perfusion.
- Finally epithelial cells resurface the injury.
- Epithelialization happens faster when wounds are kept moist and hydrated.
- Generally, when occlusive or semi-occlusive dressings are applied within 48 hours after injury, they will maintain correct tissue humidity to optimize epithelialization.
Maturation Phase (Remodelling Stage)[edit | edit source]
- Collagen is remodelled from type III to type I and the wound fully closes.
- The cells that had been used to repair the wound but which are no longer needed are removed by apoptosis, or programmed cell death.
- The collagen laid down during the proliferative phase, it is disorganized and the wound is thick.
- Collagen is remodelled into a more organized structure along lines of stress, thereby increasing the tensile strength of the healing tissues. Fibroblasts secrete matrix metalloproteinases. The enzymes facilitate remodelling of type III collagen to type I collagen[5].
- Generally, remodelling begins about 21 days after an injury and can continue for a year or more.
- Even with cross-linking, healed wound areas continue to be weaker than uninjured skin, generally only having 80% of the tensile strength of unwounded skin[6].
Clinical Significance[edit | edit source]
Any disruption in wound healing phases leads to excessive wound healing or chronic wound formation.
Excessive Wound Healing[edit | edit source]
- The pathogenesis of the excessive wound healing is not fully understood.
- An abnormal form of a wound healing that is characterized by a continuous localized inflammation.
- Excessive collagen synthesis, abnormal collagen turnover and exaggerated ECM accumulation in these wounds. e.g. "Keloid" and "hypertrophic scars".
Chronic Wound Formation[edit | edit source]
- A wound that has failed to heal in 4 weeks is defined as a chronic wound.
- Risk Factors; Age, immune status, malnutrition, infection, insufficient oxygenation or perfusion, smoking, diseases, medications, radiation, and chemotherapy are the main risk factors.
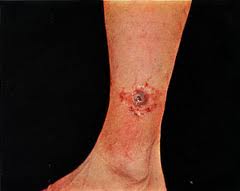
- Chronic wounds are usually classified as vascular ulcers (venous or arterial ulcers), diabetic ulcers, and pressure ulcers.[3] See image.
Other Complications can include:
- Deficient scar formation.
- Exuberant granulation.
- Deficient contraction (in skin grafts) or excessive contraction (in burns).
- Others: Dystrophic calcification[7], pigmentary changes[8], painful scars, incisional hernia.[9]
Factors Affecting the Wound Healing[edit | edit source]
- Main Risk factors are: age, immune status, malnutrition, infection, insufficient oxygenation or perfusion, smoking, diabetes, metabolic diseases, medications, radiation, and chemotherapy[3].
Also, consider:
- Extrinsic factors which include: support surfaces, friction, and shear and effective repositioning schedules.
- Local factors: moisture (keeping a wound moist improves healing)[10]; edema; faulty technique of wound closure; Ischemia and necrosis; foreign bodies[11].
Wound Care in Physiotherapy[edit | edit source]
The most common wounds that are treated by wound care physical therapist are:[12]
- Necrotic wounds.
- Stage III, IV or unstageable pressure ulcers.
- Diabetic wounds, see image.
- Chronic wounds.
- Venous and/or arterial wounds.
- Extremity wounds with oedema.
- Non-healing surgical wounds.
Physiotherapy Role[edit | edit source]
Physical therapy wound care begins with a comprehensive evaluation and the development of an individualized care plan. [12]
Common treatment approaches can include:
- Measurement and documentation of the wound characteristics.
- Cleaning of the wound.
- Debridement (removal) of any dead tissue.
- Selection and application of wound dressing.
- Application of compression if necessary.
- Education of the patient, caregivers and/or family members regarding wound care and dressing changes.
Treatment modalities may include:
- Ultrasound mist therapy
- Electrical stimulation
- Pulsed lavage
- Whirlpool
- Negative pressure vacuum therapy
- Compression therapy.
Related Resources[edit | edit source]
References[edit | edit source]
- ↑ Overview of the integumentary system (skin) image - © Kenhub. Available from; https://www.kenhub.com/en/library/anatomy/integumentary-system
- ↑ Yousef H, Alhajj M, Sharma S, Anatomy, Skin (integument), epidermis [Internet]. 2017 [cited 24 October 2020]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470464/
- ↑ 3.0 3.1 3.2 3.3 Kangal MK, Regan JP, Wound Healing [Internet]. 2020 [cited 24 October 2020]. Available from:https://www.ncbi.nlm.nih.gov/books/NBK535406/
- ↑ Herman TF, Bordoni B. Wound Classification. StatPearls Publishing; 2022.
- ↑ 5.0 5.1 Magee DJ, Zachazewski JE, Quillen WS. Pathology and intervention of musculoskeletal disorders. Elsevier Health Sciences, 2008.
- ↑ 6.0 6.1 Wound Source. Wound Healing. Available from:https://www.woundsource.com/blog/four-stages-wound-healing (last accessed 24 October 2020).
- ↑ Enoch S, Kupitz S, Miller DR, Harding KG. Dystrophic calcification as a cause for non-healing leg ulcers. International Wound Journal. 2005; 2(2):142-7.
- ↑ Chadwick S, Heath R, Shah M. Abnormal pigmentation within cutaneous scars: a complication of wound healing. Indian journal of plastic surgery 2012; 45(2):403.
- ↑ Hope WW, Waheed A, Tuma F. Incisional hernia [Internet]. 2019. Available from:https://www.ncbi.nlm.nih.gov/books/NBK435995/
- ↑ Metzgerm S. Clinical and financial advantages of moist wound management. Home Healthcare Nurse. 2004; 22(9):586-90.
- ↑ Iconomou TG, Zuker RM, Michelow BJ. Management of major penetrating glass injuries to the upper extremities in children and adolescents. Microsurgery. 1993;14(2):91-6.
- ↑ 12.0 12.1 Cleveland Clinic. Wound care.https://my.clevelandclinic.org/departments/rehabilitation/services/wound-care (accessed 24 October 2020).