The Role of the Physiotherapist in Palliative Care for People With Lymphoedema
Introduction[edit | edit source]
Lymphoedma is a swelling in the tissue spaces that develops due to an interruption in the lymphatic system. Managing lymphoedema in patients with advanced cases in palliative care is challenging[1].
The main cause of lymphoedema in the Western world is cancer and its treatment, such as surgery to remove the lymph nodes, radiotherapy to nodal areas or local metastatic disease, or a combination of the above.
The resultant swelling will depend on which area of the body the lymphatics have been damaged in, e.g. in the treatment of Breast Cancer the nodes in the axilla may be removed or be treated by radiotherapy and may subsequently lead to lymphoedema in the arm. There are many physical, psychological and social factors that can affect the swelling and its management.
- Adopting a holistic approach to patient care as used in the palliative setting ensures a patient centred plan of care is developed.
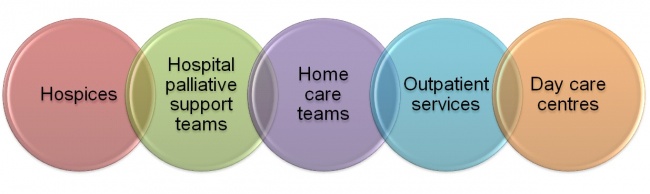
- Hospices are probably the most well known setting for palliative care, however palliative care takes place in multiple settings that people often do not think of (see image R)
- The World Health Organization (2004) defines palliative care as an approach that improves the quality of life of those patients who face life-threatening illness, and their families, through prevention and relief of suffering using early identification, assessment and treatment of pain and other problems.
Physiotherapy Management[edit | edit source]
The palliative lymphoedema assessment aims to:
- Understand the patient’s main concerns, goals and priorities
- Help the clinician understand the main cause of and the mechanisms behind the swelling
- Understand the underlying condition and how quickly it is progressing
A systematic review and meta-analyses suggest the need for complete awareness of the factors contributing to the wide variability in lymphedema in order to improve QOL people living with cancer-related lymphedema in low and middle-income countries[2].
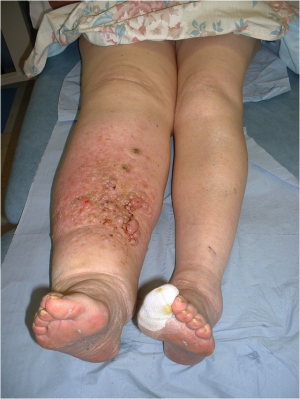
Oedema is a direct result of multiple factors relating to a terminal illness.
- It can be distressing for patients and a management challenge for health professionals. It is estimated that 5-10% of new referrals to palliative care have oedema but this is also thought to be underestimated[3].
- Heavy and swollen limbs can cause proximal pain while patients with active malignancy may experience neuropathic pain due to nerve compression.
- Up to 67% of patients experience pain as a result of oedema.
- For patients, lymphoedema may be seen as a constant reminder of their cancer or illness. The swollen limb is heavy and uncomfortable which leads to a reduction in mobility and function[4].
Assessment[edit | edit source]
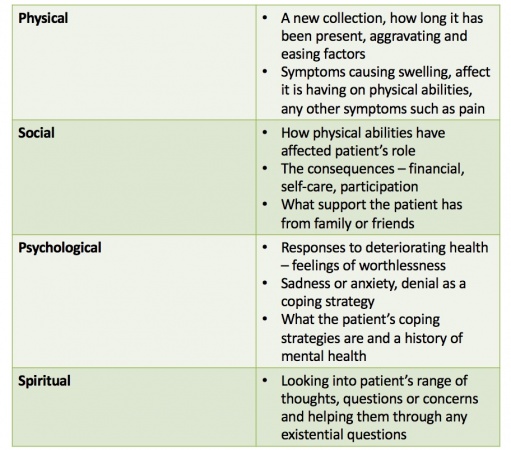
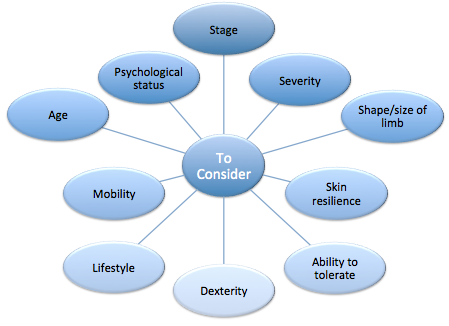
The assessment should include physical, social, psychological and special aspects along with a history of the illness and medication[3].
- An examination is then carried out and baseline measurements are taken. This helps to plan the programme of care, assess the response to treatment, identify risks of potential complications and may show signs that confirm the cause of the oedema.
- Due to the nature of the disease patients priorities often change, therefore the assessment is ongoing and reviews of priorites and goals occur regularly[4].
- From the assessment, the priority will be to negotiate a care plan based on the patient’s problems and the best approach to alleviate these issues[5].
- The assessment includes a full history of the oedema[3].
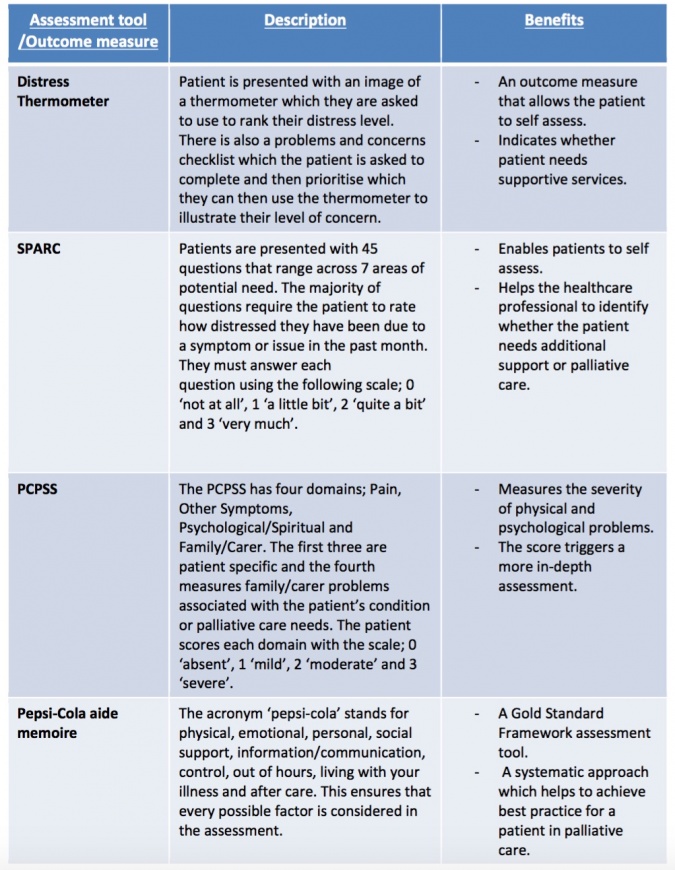
The use of an outcome measure as a tool can be very valuable during an assessment[6]:
- Ensures the focus is on the patients specific needs
- When used effectively, it provides a structured assessment conversation allowing the patient’s worries to be prioritised
- Ensures all areas of assessment are covered
- Patients become familiar with the tool and it can be used by different healthcare professionals involved in their care
The most commonly used tools within a palliative setting are shown in table at R[6]
Treatment challenges [edit | edit source]
The main aim of palliative rehabilitation is to set treatment goals that allow a patient to maintain or improve functions and delay disease progression for as long as possible.
- Losing functional ability can cause a patient to view themselves differently and reduces their independence, consequently this can impact their psychological well-being. Therefore, physiotherapists have an important role in helping patients maintain their independence and achieve other goals they may have[7]. Goals need to be short-term and adaptable due to the ever changing nature of a palliative condition. Sometimes it is necessary to carry out a new assessment each time you see a patient due to this varying nature[7].
- In a palliative setting goal setting must be sensible and specific to the patient's individual needs. Goal setting with the patient should include close interaction with the physiotherapist and other health professionals involved in their care to ensure they are adequately educated in a range of self-management strategies. Acceptable palliative goals include, slowing down the progression of swelling and reducing other symptoms associated with lymphoedema [3].
- Patients may have dramatically reduced physical capacity. If their illness has compromised neurologic structures, they may be plegic or paretic, and therefore unable to self- bandage or perform remedial exercises. Further, they may be significantly limited by symptoms such as fatigue and dyspnoea due to marked deconditioning. The coordination, dexterity and strength requirements for bandaging and donning compression garments can elude even healthy patients; therefore, these potential difficulties should be considered when formulating a management plan for terminally ill patients [3].
Physiotherapy Interventions[edit | edit source]
Complete Decongestive Therapy (CDT)
Traditionally lymphoedema is managed with CDT and is based on four pillars of care: compression, massage, skin care and exercise[8]. Standard CDT may not be appropriate due to weakness and frailty so this must be modified and adapted[5]. Modified CDT has the capacity to significantly benefit patients with far advanced disease who have lymphoedema or multi-factorial oedema. CDT can enhance patients’ function and comfort while preventing needless complications and enhancing psychological well-being [3].
Standard CDT
- Goals to reduce swelling, transition to garments, life-long maintenance, self-care
- Four elements of CDT
- Two distinct phases of treatment
- Definitive contraindications to treatment
Palliative CDT
- Goals to provide comfort, support, relief of symptoms, maintain function, include caregivers in care
- CDT elements may be modified or omitted
- Less distinction between phases of treatment
- Contraindications now relative[9]
Compression Therapy
Adapting multilayer bandaging (palliative bandaging)
- In general, due to patients’ inability to tolerate compression, palliative bandaging will need to be gentler.
- Compression pressure may need to be reduced or more gradually increased compared to standard treatment.
- Be especially aware of fragile skin.
- Check the skin frequently and even in the best circumstances it is not wise to leave bandages on for more than 48 hours.
- In cases of decreased sensation or numbness, compression therapy may be contraindicated, or if used, be very closely monitored.
- Wounds and lymphorrhoea are common in end-of-life care. One may need to incorporate absorbent dressings to deal with this.
Below are listed main points to be aware of re standard vs palliative bandaging
Standard bandaging
- Full standard pressure
- Multilayer bandages
- 24-hour bandaging during intensive phase
- Foam padding used
- Transition to compression garments
Palliative bandaging
- Reduced pressure
- Fewer layers used
- Intensive treatment for lymphorrhoea may require frequent re-application of bandages
- Soft padding generally better tolerated
- May transition to lighter support bandaging, or to compression garments or continue palliative bandaging[9]
Compression Garments/Hosiery
Use of compression garments
- Donning aids can be helpful to protect fragile skin.
- As limb size may vary from day to day in the palliative patient, fitted compression garments are often not suitable or well-tolerated and can cause dangerous constriction.
- The adaptable system of multilayered compression bandaging is often a better alternative than garments.
- Nighttime, non-elastic compression foam devices (with or without Velcro closures) may be more flexible and easier for patients or caregivers to apply.
- Light compression gloves, ‘power sleeve’ inner foam inserts (from night compression devices) can also be used as light support.
- In the case of gynaecological and genital oedema, light compression garments such as bicycle shorts, or purpose-made lycra shorts, will supply some compression[9].
- Garments are either constructed as a flat knit or a round knit. Flat knit are knitted as a flat garment then joined at the seams, these are generally thicker and firmer. Round knitted garments are viewed as more aesthetically pleasing, as they are thinner than flat knit and are continuously knitted cylindrically without any seams[8].
- Once measurement and construction of the garment is completed, the health professional will assess the garment for fit and check that the patient or carer is able to apply and remove the garment correctly. Advice regarding the care at home will be provided in person and leaflets may also be given to the patient[8]. If garments are poorly fitted, the swelling may not be contained and damage can occur to the tissues. This could result in discomfort and reduced tolerance leading to patients being unwilling to use compression hosiery as a long-term management option[10]. As well as limb compression garments, if patients have trunk or breast lymphoedema garments or specialised bras can be provided[8].
Skin Care
In palliative patients, the skin can become very fragile and the aim is to prevent any damage.
- As a consequence of swelling, large skin folds can appear where infections may develop [8].
- Infections can also arise if the skin becomes damaged or broken, therefore adequate skin care to maintain the integrity and manage any problems that occur is fundamental in the care of people with lymphoedema[8][11].
- Care should be taken to wash and dry thoroughly, especially between the digits and any skin folds. Apply an unscented moisturiser to prevent drying of the skin. If compression hosiery is being applied care should be taken to prevent damage during application.
- Any breaks in the skin can result in lymphorrhoea – lymph fluid leaking onto the surface of the skin. The extent of the lymphorrhoea will depend on the size of the tear in the skin and whether the limb is dependent or not. The fluid causes maceration of the skin, soaking of clothing, footwear and bedding and can be very cold and distressing for the patient.
- Mild lymphorrhoea can be treated with an absorbent dressing and continuing with compression hosiery. In some cases, however, the management is purely palliative as bandaging may not stop the lymphorrhoea, especially if the patient is sitting in a chair for the majority of the time. Bed rest is occasionally the only cure for lymphorrhoea and this inevitably takes place as the patient gets closer to death[5]
Exercise
Based on new evidence, traditional beliefs about the effects of exercise on lymphoedema have been disproven.
- MacMillan Cancer Support[12] state that many individuals experience reduced quality of life following cancer treatment due to secondary complications, which can include lymphoedema.
- Despite the general well known benefits of exercise including reduced risk of chronic diseases and the positive impact it can have on mental [13], studies report that cancer survivors often fail to return to their pre-diagnosis levels of physical activity[14][15].
Traditionally strenuous exercise was discouraged in patients with lymphoedema based on the belief that it may exacerbate the condition[16]. However recent studies and systematic reviews contradict this statement. Cancer Research UK provides an informative section on their website for people with lymphoedema.
- Normal use of the limb can be sufficient to assist lymphatic flow but even normal use may be restricted in palliative patients.
- Pain, weight of the limb, fatigue or neurological impairment will impact limb mobility.
- Relatives and carers could assist in some passive exercises if tolerated.
- Elevation of the limb will help reduce the gravitational component of the swelling, e.g. supporting the arm on a pillow or cushion to prevent pooling at the
- Aerobic activities to stimulate lymphatic return should be encouraged within the patients tolerance. Aerobic exercise minimises psychological distress and fatigue among cancer patients, even those with advanced and widely spread disease[3].
The following table summarises some of the studies that have been conducted surrounding this topic.
| Study Title | Design | Participants | Intervention | Outcome Measures | Results |
|---|---|---|---|---|---|
| Effect of Upper Extremity Exercise on Secondary Lymphedema in Breast Cancer Patients: A Pilot Study[17] | Pilot study | 14 breast cancer survivors with unilateral upper limb lymphoedema | Progressive upper body exercise programme – 8 weeks vs. no intervention | Arm circumference and volume Medical Outcomes Trust Short From 36 – survey |
No changes in lymphoedema Physical, general and vitality components of QOL improved |
| Exercise and Secondary Lymphedema: Safety, Potential Benefits, and Research Issues[18] | RCT | 32 breast cancer survivors with lymphoedema | Mixed type exercise programme – 12 weeks vs. no intervention | Bioimpedence spectroscopy and perometry | No changes – did not exacerbate lymphoedema |
| Weight lifting in Women with Breast-Cancer-Related Lymphoedema[19] | RCT | 141 breast cancer survivors with stable arm lymphoedema | 2x weekly progressive weight lifting vs. no intervention | Severity of lymphoedema, number and Incidence of exacerbations and symptoms and muscle strength | No effect on limb swelling, reduced exacerbations and symptoms, increased muscle strength |
| Systematic Review and Meta-Analysis of the Effects of Exercise for Those With Cancer-Related Lymphoedema[20] | Systematic review and meta-analysis | Individuals with secondary lymphoedema 25 studies included |
Range of exercises | Lymphoedema and associated symptoms | No effect on lymphoedema or associated symptoms Insufficient evidence for use of compression during exercise |
| Exercise in patients with lymphedema: a systematic review of the contemporary literature[21] | Systematic review | Cancer patients with or at risk of lymphoedema 19 studies included |
Range of exercises | Lymphoedema development and exacerbations | No development or exacerbation of lymphoedema |
| Weight training is not harmful for women with breast cancer-related lymphoedema: a systematic review[22] | Systematic review and meta-analysis | Women with or at risk of developing breast cancer related lymphoedema 11 studies included |
Progressive weight training exercise | Severity and incidence of arm lymphoedema, upper limb muscle strength, BMI and QOL | Increased strength without affecting arm volume or incidence of lymphoedema No effect on BMI Potential to improve some elements of QOL |
- The studies in the table above used a range of different exercise modes, there is little evidence to recommend the optimal mode of exercise for people with lymphoedema; therefore larger scale trials are required to evaluate this. However, the most recent systematic review discussed that by giving people a choice over their exercise method promoted adherence to the programme[22].
- Singh et al.[20] also reviewed the use of compression garments during exercise. Unfortunately due to the range of effects that wearing compression can have during exercise, there was no definitive answer to whether compression garments should be worn or not. The authors suggest that this decision should be made on an individual basis considering factors such as stage, severity, and stability of lymphoedema and patient preference.
- Evidence suggests that exercise supervised by a qualified professional i.e. a physiotherapist in the first instance - this will ensure correct technique and reduce injury risk[21]. It can be concluded from the body of evidence including large randomised controlled trials and recent systematic reviews that strenuous training, as previously thought does not lead to the development or worsening of lymphoedema. Despite showing little effect on lymphoedema, it is important to note the benefits that exercise has on individual’s strength, functioning and quality of life. These benefits outweigh any risks that were previously suggested.
Manual Lymphatic Drainage (MLD)
Manual lymphatic drainage (MLD) is highly effective in the palliative setting. It has pain relieving properties and can significantly clear even tight, malignant oedema. MLD should avoid areas of dermal compromise, cancerous invasion, extensive fibrosis, or skin hypersensitivity. However, if MLD has been found to be effective over such areas, the treatment should be continued as the main aim of palliative care is to better a patient’s comfort and quality of life[3].
see Complete Decongestive therapy for more information
Additional Resources[edit | edit source]
References[edit | edit source]
- ↑ Beck M, Wanchai A, Stewart BR, Cormier JN, Armer JM. Palliative care for cancer-related lymphedema: a systematic review. Journal of palliative medicine. 2012 Jul 1;15(7):821-7.Available from:https://www.researchgate.net/publication/225183804_Palliative_Care_for_Cancer-Related_Lymphedema_A_Systematic_Review (last accessed 21.8.2020)
- ↑ Torgbenu E, Luckett T, Buhagiar MA, Chang S, Phillips JL. Prevalence and incidence of cancer related lymphedema in low and middle-income countries: A systematic review and meta-analysis.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 ILF. The management of lymphoedema in advanced cancer and oedema at the end of life. http://www.lympho.org/mod_turbolead/upload/file/Palliative%20Document%20-%20protected.pdf (accessed 11 October 2015).
- ↑ 4.0 4.1 Todd M. Understanding lymphoedema in advanced disease in a palliative care setting. International Journal of Palliative Nursing 2009;15(Suppl 10):474-480. http://www.ncbi.nlm.nih.gov/pubmed/20081719 (accessed 17 December 2015).
- ↑ 5.0 5.1 5.2 Todd M. Managing lymphoedema in palliative care patients. British journal of nursing 2009;8(Suppl 8):466-472. http://www.researchgate.net/profile/Marie_Todd/publication/24308357_Managing_lymphoedema_in_palliative_care_patients/links/0c96052a1aded1e3a3000000.pdf (accessed 17 December 2015).
- ↑ 6.0 6.1 National Cancer Action Team. Holistic needs assessment for people with cancer. http://www.ncsi.org.uk/wp-content/uploads/Holistic-Needs-Assessment-practical-guide.pdf (accessed 11 December 2015).
- ↑ 7.0 7.1 Frymark U, Hallgren L, Reisberg A. Physiotherapy in palliative care – a clinical handbook. Sweden: Stockholms Sjukhem, 2009.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 Lymphoedema Framework. Best Practice for the Management of Lymphoedema. International consensus. http://www.woundsinternational.com/media/issues/210/files/content_175.pdf (accessed 15 January 2016).
- ↑ 9.0 9.1 9.2 Towers A, Hodgson P, Shay C, Keeley V. Care of palliative patients with cancer-related lymphoedema. J Lymphoedema. 2010;5(1):72-80.Available from:https://www.woundsinternational.com/uploads/resources/content_11200.pdf (last accessed 21.8.2020)
- ↑ Doherty D, Morgan P, Moffatt C. Hosiery in lower limb lymphoedema. Journal of Lymphoedema 2009;4(Suppl 1):30-37. http://www.journaloflymphoedema.com/ (accessed 15 January 2016).
- ↑ Wigg J, Lee N. Managing lymphoedema and chronic oedema. Nursing and Residential Care 2015;17(Suppl 4):192-197. http://www.magonlinelibrary.com/doi/10.12968/nrec.2015.17.4.192 (accessed 15 January 2016).
- ↑ MacMillan Cancer Support. Cured – but at what cost? http://www.macmillan.org.uk/Documents/AboutUs/Newsroom/Consequences_of_Treatment_June2013.pdf (accessed 16 January 2016).
- ↑ NHS Choices. Benefits of Exercise. http://www.nhs.uk/Livewell/fitness/Pages/Whybeactive.aspx (accessed 16 January 2016).
- ↑ Irwin ML, Crumley D, McTieran A, Bernstein L, Baumgartner R, Gilliand F, Kriska A, Ballard-Barbash R. Physical Activity Levels before and after a Diagnosis of Breast Carcinoma, The Health, Eating, Activity and Lifestyle (HEAL) Study. Cancer 2003;97(Suppl 7):1746-1757. http://www.ncbi.nlm.nih.gov/pubmed/12655532 (accessed 15 January 2016).
- ↑ Harrison S, Hayes SC, Newman B. Level of physical activity and characteristics associated with change following breast cancer diagnosis and treatment. Psycho-Oncology 2009;18:387-394. http://onlinelibrary.wiley.com/doi/10.1002/pon.1504/references (accessed 15 January 2016).
- ↑ Cheifetz O, Haley L. Management of secondary lymphoedema related breast cancer. Canadian Family Physician 2010;56:1277-1284. http://www.ncbi.nlm.nih.gov/pubmed/21375063 (accessed 15 January 2016).
- ↑ McKenzie DC, Kalda AL. Effect of Upper Extremity Exercise on Secondary Lymphedema in Breast Cancer Patients: A Pilot Study. Journal of Clinical Oncology 2003;21(Suppl 3):463-466. http://www.ncbi.nlm.nih.gov/pubmed/12560436 (accessed 15 January 2016).
- ↑ Hayes SC, Reul-Hirche H, Turner J. Exercise and Secondary Lymphedema: Safety, Potential Benefits, and Research Issues. Medicine and Science in Sports and Exercise 2009;483-489. http://www.ncbi.nlm.nih.gov/pubmed/19117320 (accessed 15 January 2016).
- ↑ Schmitz KH, Ahmed RL, Troxel A, Cheville A, Smith R, Lewis-Grant L, Bryan CJ, Williams-Smith CT, Greene QP. Weight lifting in Women with Breast-Cancer-Related Lymphoedema. The New England Journal of Medicine 2009;361:664-673. http://www.ncbi.nlm.nih.gov/pubmed/19675330 (accessed 15 January 2016).
- ↑ 20.0 20.1 Singh B, Disipio T, Peake J, Hayes SC. Systematic Review and Meta-Analysis of the Effects of Exercise for Those With Cancer-Related Lymphoedema. Archives of Physical Medicine and Rehabilitation 2015;1-30. http://www.ncbi.nlm.nih.gov/pubmed/26440777 (accessed 15 January 2016).
- ↑ 21.0 21.1 Kwan M, Cohn JC, Armer JM, Stewart BR, Corimer N. Exercise in patients with lymphedema: a systematic review of the contemporary literature. Journal of Cancer Survivorship 2011;5:320-336. http://www.ncbi.nlm.nih.gov/pubmed/22002586 (accessed 15 January 2016).
- ↑ 22.0 22.1 Paramanandam VS, Roberts D. Weight training is not harmful for women with breast cancer-related lymphoedema: a systematic review. Journal of Physiotherapy 2014;60:136-143. http://www.ncbi.nlm.nih.gov/pubmed/25086730 (accessed 15 January 2016).