Achilles Tendinopathy
Original Editor - Karolyn Conolty
Top Contributors - Aline Tréfois, Admin, Puja Gaikwad, Kim Jackson, Eva Roose1, Rachael Lowe, Lucinda hampton, Laura Ritchie, Shauni Van Overstraeten, Karolyn Conolty, Chi Ngai LO, Christopher, Fasuba Ayobami, Simisola Ajeyalemi, Birgit Schauvliege, Wanda van Niekerk, Naomi O'Reilly, Camille Linussio, Olajumoke Ogunleye, 127.0.0.1, Vidya Acharya, Rucha Gadgil, Eric Robertson, Samuel Adedigba, Jess Bell, Khloud Shreif and David Bayard
Introduction[edit | edit source]
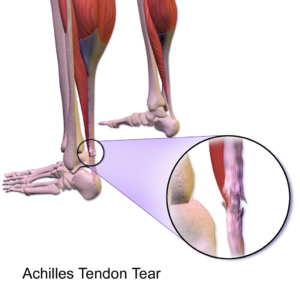
Achilles tendinopathy (common overuse injury) refers to a combination of pathological changes affecting the Achilles tendon usually due to overuse and excessive chronic stress upon the tendon. It can be seen both in athletes and non-athletes. It may or may not be associated with an Achilles tendon tear. A lack of flexibility or a stiff Achilles tendon can increase the risk of these injuries.[1][2]
Surgical specimens show a range of degenerative changes of the affected tendon, eg the tendon fiber structure and arrangement; an increase in glycosaminoglycans (water-binding molecules that can hold nearly 1,000 times their own weight), which may explain the swelling of the tendon. [3][4]
The precise cause of tendonitis remains unclear. Even though tendonitis of the Achilles tendon is often connected to sports activities, the ailment is also often found in people who do not practice sports. The biggest cause is the excessive overburdening of the tendon. A light degeneration of the Achilles tendon can be latently present, but the pain only appears when the tendon is overburdened. It is also noted that the ailment is usually not preceded by trauma[5][6].
Terminology[edit | edit source]
The current term that is recommended to describe this cohort of patients is ‘tendinopathy’. Cook and Purdum[7]proposed a new strategy when approaching tendon pain, and this is called the Tendon Continuum. The continuum model proposed a model for staging tendinopathy based on the changes and distribution of disorganization within the tendon. Three stages are as follows:
- Reactive tendinopathy
- Tendon dysrepair
- Degenerative tendinopathy
It has been suggested that the tendon can move up and down this continuum and this can be achieved through adding or removing load to the tendon especially in the early stages of tendinopathy.
Location[edit | edit source]
Achilles tendinopathy can be described as an insertional or mid-portion, the difference is in the localization. The insertional form is situated at the level of transition between the Achilles tendon and the bone (<2 cm from its insertion), the midportion form is located at the level of the tendon body (>2 cm from its insertion).[2]
Clinically Relevant Anatomy[edit | edit source]
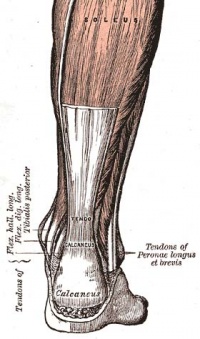
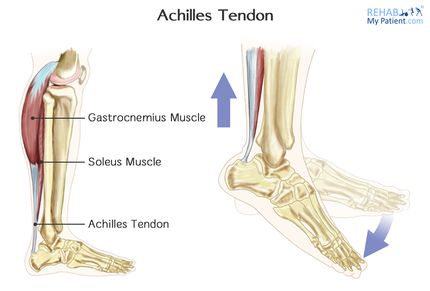
The Achilles tendon is the biggest and strongest tendon in the human body. The tendon has the capacity to resist large tensile forces. It stems from a distal joining of the gastrocnemius and soleus muscle and inserts at the bottom of the calcaneus.
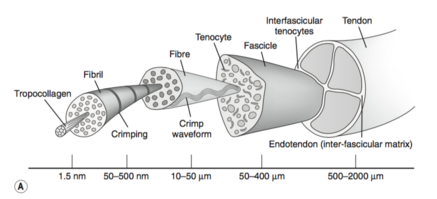
A typical tendon structure consists of thin, cylindrical cells and an extracellular matrix. The cells of the tendon (the tenocytes and tenoblasts) are responsible for the synthesis of all of the components of the extracellular matrix. Inside the matrix, we find bundles of type I collagen and elastin. This type-I collagen is responsible for the strength of the tendon. Between the collagen, there is a ground substance, which is made up of proteoglycans and glycosaminoglycans. [8]
The Achilles tendon is surrounded by paratenon which works as an elastic sleeve around the tendon which allows the tendon to move freely between surrounding tissue. The paratenon, membrane-like areolar structure consisting of loose connective tissue, is found around extraarticular tendons without a synovial sheath (eg the Achilles tendon). It supplies the tendon with blood from nearby vessels at several levels[9][10]
The blood supply throughout the length of the tendon is poor, as shown by the small numbers of blood vessels per cross-sectional area, especially the region 4-6cm above the calcaneus. Poor vascularity may characterize a slow healing rate following trauma[11][12].
Aetiology[edit | edit source]
A reactive tendon is the 1st stage on the tendon continuum and is a non-inflammatory proliferative response in the cell matrix. This is as a result of compressive or tensile overload. Straining the tendon during physical exercise has been seen as one of the biggest pathological stimuli and systematic overloading of the Achilles tendon above the physiological limit can cause a micro-trauma. Repetitive micro-traumas that are linked with a non-uniform tension between the gastrocnemius and soleus, cause frictional forces between the fibres and abnormal concentrations of the loading in the Achilles tendon. This has consequences such as the inflammation of the tendon sheath, degeneration, or a combination of both. Without the minimum time for recovery, this can lead to tendinopathy [13].
Decreased arterial blood flow, local hypoxia, decreased metabolic activity, nutrition, and persistent inflammatory response have been suggested as possible factors that could lead to chronic tendon overuse injuries and tendon degeneration.
The most common and perhaps the most important malalignment is one of the ankles caused by overpronation of the foot. Increased foot pronation has been proposed to be associated with Achilles tendinopathy.
- In acute trauma, the external factors dominate, while injuries caused by overuse generally have a multifactorial origin. The acute phase of Achilles tendinopathy is caused by acute overload, blunt trauma, or acute muscle fatigue, and is characterized by an inflammatory reaction and edema formation. If the treatment of the acute phase fails or if they overlooked it, it can cause a fibrin and form adhesions off the tendon.
- The progression of the reactive tendinopathy to tendon disrepair can occur if the tendon is not offloaded and allowed to regress back to the normal state. During this phase, there is the continuation of increased protein production which has been shown to result in separation of the collagen and disorganization within the cell matrix. This is the attempt of tendon healing as with the 1st phase but with greater involvement and breakdown physiologically.
- Degenerative tendinopathy is the final stage on the continuum and it is suggested that at this stage there is a poor prognosis for the tendon and changes are now irreversible. Often, tendon degeneration is found in combination with peri-tendinous adhesions, but this does not mean that one condition causes the other one.
Recent research also shows
- Older age, higher android fat mass ratio, and waist circumference > 83cm, in men is associated with a higher chance of having Achilles Tendinopathy[14][15].
- The presence of the COL5A1 gene variant was also found to be a possible risk factor. This gene is normally responsible for the production of tendon protein, but patients with the condition were shown to have significantly different allele frequencies of the COL5A1 BstUI RFLP compared with normal subjects[16][17][18][19]
- Individuals with chronic Achilles tendinopathy showed signs of both peripheral and central sensitization to pain.[20] Physical therapy management of chronic Achilles tendinopathy should factor in this point in interventions used to treat chronic tendinopathies.
In summary, the effects of overuse, poor circulation, lack of flexibility, gender, endocrine, or metabolic factors can lead to tendinopathies. The structure of the tendon is disrupted by this repetitive strain (often eccentric in nature), and collagen fibers begin to glide past one another, breaking their cross-links and causing tissue denaturation, leading to inflammation. This accumulating microtrauma is thought to weaken collagen cross-linking and affect the non collagenous matrix and the vascular elements of the tendon, finally leading to tendinopathy.[6]
Risk factors[edit | edit source]
There are some known risk factors related to the pathology of Achilles tendinopathy. few of these risk factors include:
- Obesity
- High blood pressure
- Rapid changed to load
- Type II Diabetes
- Prolonged steroid use
- Family history of tendinopathy
- Other factors include inappropriate footwear and the elderly population.
Clinical Presentation[edit | edit source]
Morning pain is a hallmark symptom because the Achilles tendon must tolerate a full range of movement including stretch immediately after getting up in the morning. Symptoms are typically localized to the tendon and the immediate surrounding area.
Swelling and pain are less common. The tendon can appear to have subtle changes in outline, becoming thicker in the A-P and M-L planes.[21]
With people who have tendinopathy of the Achilles tendon that has a sensitive zone, combined with intra-tendinous swelling, that moves along with the tendon and of which sensitivity increases or decreases when the tendon is put under pressure, there will be a high predictive value that in this situation there is a case of tendinosis. [22] The affected side of the tendon shows a larger diameter, higher stiffness, and lower strain in comparison to the non-affected side.
Differential Diagnosis[edit | edit source]
- Plantar fasciitis
- Calcaneal fracture stress
- Heel pad syndrome (deep, bruises and pain in the centre of the heel)
- Haglund deformity - this is a prominence of the calcaneus that can cause bursitis between the calcaneus and the Achilles tendon
- Sever's Disease - Irritation of the back of the calcaneus at the growth plate. Occurs in young children and adolescents, particularly around puberty and during growth spurts.
- Posterior Ankle Impingement
- Medial Tendinopathy
- Retrocalcaneal Bursitis
- Sural Nerve
- Lumbar Radiculopathy
- Ankle OA
- Deep vein thrombosis
- Partial Achilles Tendon Rupture. [21]
Examination[edit | edit source]
Subjective Assessment: Important for providing clues related to the mechanism of injury and history of the condition. Clinicians can use a subjective report of pain located 2 to 6 cm proximal to the Achilles tendon insertion that began gradually and pain on palpation to diagnose Achilles tendinopathy[23].
Objective examination: Important to fully assess the lower limb. Assessing the hip and knee will give clues to biomechanical contributions and muscle imbalances. In the foot and ankle we are looking for more local contributory and resulting factors:
- Observation: looking for muscle atrophy, swelling, asymmetry, joint effusions, and erythema. Atrophy is an important clue to the duration of the tendinopathy and it is often present with chronic conditions. Swelling, asymmetry, and erythema in pathologic tendons are often observed in the examination. Joint effusions are uncommon with tendinopathy and suggest the possibility of intra-articular pathology.
- Range of motion testing, strength, and flexibility: often limited on the side of the tendinopathy[24][22].
- Palpation: tends to elicit well-localized tenderness that is similar in quality and location to the pain experienced during activity[25] Palpation often reveal palpable nodules and thickening.
- Anatomic deformities: eg forefoot, heel varus, excessive pes planus, or foot pronation, should receive special attention. These anatomic deformities are often associated with this problem[5][26]
- A positive arc sign, and positive findings on the Royal London Hospital test[23]Shown in 30 second videos below.
Clinicians should use physical performance measures, including hop and heel-raise endurance tests, as appropriate, to assess a patient’s functional status and document findings.
When evaluating physical impairment over an episode of care for those with Achilles tendinopathy, one should measure ankle dorsiflexion range of motion, subtalar joint range of motion, plantar flexion strength and endurance, static arch height, forefoot alignment, and pain with palpation[23]
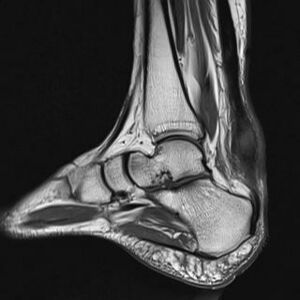
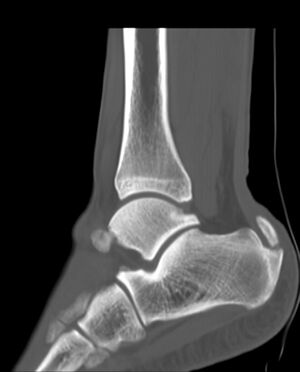
Imaging studies: not necessary to diagnose Achilles tendonitis but may be useful for differential diagnosis. Ultrasound is the imaging modality of the first choice as it provides a clear indication of the tendons' width, changes of water content within the tendon and collagen integrity, as well as bursal swelling. An MRI may be indicated if the diagnosis is unclear or if symptoms are atypical. The MRI may show an increased signal within the Achilles.[21] The use of non-invasive image-forming methods for the assessment of the Achilles tendon (mechanical, structural, and biomechanical characteristics) in vivo is relatively young. Ultrasound elastography and ultra-high-field magnetic resonance imaging (MRI UHF) have recently emerged as potential powerful techniques to examine the tendon tissues. [29]
Outcome Measures[edit | edit source]
Patient-reported outcome measures such as:
- A global measure of lower extremity function: e.g., The Lower Extremity Functional Scale (LEFS) ‐ not specific to Achilles tendinopathy
- A detailed questionnaire, specific to Achilles tendinopathy e.g. the VISA‐A questionnaire.[21][30]
Patient-specific functional outcome measure such as:
- How much weight can be applied to the plantarflexed foot on a weighing scale before the onset of pain
- The number of heel raises before the onset of pain
- The number of heel drops before the onset of pain
- The number of heel drops with a specific weight in a backpack before the onset of pain
- How far can the client walk or run before the onset of pain
Recently, it was suggested that clinicians should use the Victorian Institute of Sport Assessment-Achilles (VISA-A) to assess pain and stiffness, and either the Foot and Ankle Ability Measure (FAAM) or the Lower Extremity Functional Scale (LEFS) to assess activity and participation in patients with a diagnosis Achilles tendinopathy.[23]
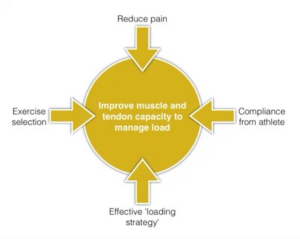
Physiotherapy Management[edit | edit source]
It is good to educate your patients on activity modification and counsel them appropriately. For patients with nonacute Achilles tendinopathy, clinicians should advise that complete rest is not indicated and that they should continue with their recreational activity within their pain tolerance while participating in rehabilitation. Clinicians may counsel patients with Achilles tendinopathy. Key elements of patient counselling could include[23];
- Theories supporting the use of physical therapy and the role of mechanical loading
- Modifiable risk factors, including body mass index and shoe wear
- Typical time course for recovery from symptoms.
The Achilles Tendinopathy Toolkit is an evidence-based clinical decision making aid to assist clinicians in their management of Achilles tendinopathy. See infographic below.
.
Optimize Biomechanics[edit | edit source]
Individuals presenting with Achilles tendinopathy should have a full biomechanical assessment. The BC Physical Therapy Tendinopathy Task[31] Force suggests there is a small amount of clinical evidence to support the use of orthotics in the acute stage and a moderate amount of clinical evidence to support the use of orthotics in the chronic stage. Clinically consider using orthotics, perhaps using taping first, in the acute stage; consider using orthotics in the chronic stage.
Controlled Tendon Loading[edit | edit source]
Less burdening activities should be encouraged so that the burden on the tendon decreases, however, complete immobilization should be avoided since it can cause atrophy[32][5] The BC Physical Therapy Tendinopathy Task Force[31] suggests that there is a large amount of clinical evidence to support the use of exercise in the chronic stage but the precise parameters to ensure effectiveness is not clear. Eccentric exercise, in particular, is supported although some protocols use both concentric and eccentric exercises. One RCT showed heavy slow resistance training is equally as effective as eccentric training.
It has been shown that strength training, that is stimulated externally and is linked to functional tasks, not only helps reduce tendon pain but modulate excitatory and inhibitory control of muscle, and thus potentially tendon load [33]. A popular and effective option is eccentric strength training.[5] In the past decade, eccentric exercises have been shown to have positive effects on Achilles tendinopathy and became the main non-surgical choice of treatment for Achilles tendinopathy[34]
There is no convincing evidence that the most effective exercise regimen.
- A recent systematic review concluded that there is little clinical and mechanistic evidence that supports the use of the eccentric component and compares that well-conducted studies of different load programs are largely lacking[34].
- New loading based exercise regimes such as isolated concentric exercise, heavy slow resistance training (HSR), and eccentric-concentric have more recently been proposed but lack solid scientific evidence for their effectiveness in Achilles tendinopathy[35].
Thorough management guidelines for Achilles tendinopathy is covered in detail in the Achilles Tendinopathy Toolkit.
The key goal of tendinopathy treatment is to improve the energy storage capacity of the tendon. It is the ability for the tendon and associated muscle to work and manage load, essentially acting as a ‘spring’ in storing and then releasing energy. Three key exercises for Achilles tendinopathy are:
- Isometric Loading
- Isotonic Loading
- Energy Storage Loading. [36]
Achilles Tendinopathy Toolkit: Section D - Exercise Programs is a great place to look for exercise appropriate programs
Phase 1: Isometric Loading- Achilles tendon holds[edit | edit source]
Management of Achilles tendon pain has changed a lot in recent years. One significant change is the emergence of isometric tendon loading as a mainstay of tendinopathy treatment. Isometric tendon loading has been found to have pain-relieving effects on tendons, while simultaneously maintaining some baseline strength. Depending on symptoms and tendon irritability these can be performed with either double legs or a single leg. For highly irritable (reactive) Achilles tendons double leg holds, of often shorter duration, and fewer repetitions may be performed. The position of the isometric hold can either be mid or end of the range (ie right up on the toes, or halfway up).
Phase 2: Isotonic Loading- Calf raises[edit | edit source]
These exercises are often commenced once the athlete's pain level and the tendon’s irritability reduces. There are no ‘hard and fast’ rules for when to start an athlete on isotonic loading for the rehabilitation of Achilles tendinopathy. Graduated isotonic loading is initiated once they have less than 5/10 pain on NRS or tolerable and acceptable pain on repeated single leg calf raises, and their morning tendon stiffness has been reduced significantly.
The ultimate goal of the isotonic exercise is to develop strength in the tendon and the surrounding muscles. In the case of the Achilles tendon, the strength of soleus and gastrocnemius muscles plays a major role. Recurring loading, such as when walking or running does not induce enough adaptation of the tendon matrix, nor the work capacity of the muscle-tendon unit. Hence heavier loads are required with the isotonic loaded exercises.
- Isotonic seated calf raises can be performed with a gradual increase in loading. Perform each repetition of 3-6 seconds in length to develop tension in the tendon.
- Isotonic standing calf raises should be performed at the mid-range of the muscle’s movement. The benefit of performing Heavy slow resistance (HSR) exercise in the mid-range is that it will avoid the compression of the tendon at end of the range that can occur with exercising with heavier loads. For example, at the very end range of ankle plantarflexion (toes pointed), or dorsiflexion (think letting the heel drop off the edge of a step) the Achilles tendon is subject to compressive loads against the heel bone (calcaneum) which can be potentially irritating and pain-producing.
Phase 3: Energy Storage Loading- Plyometric Exercises[edit | edit source]
The crucial last stage of rehabilitation is the initiation and execution of ‘energy storage’ tendon exercises. These exercises include deformation of the tendon with jumping and hopping based exercises. These exercises help the tendon to regain its capacity to absorb and then release energy via the stretch-shortening cycle, that happens when an athlete lands and then pushes off at toe-off.
These exercises can be initiated when the athlete is reporting minimal or markedly reduction in morning stiffness in the Achilles tendon on waking. In addition, other criteria for starting an athlete on these exercises include: when the athlete has been progressing well with isotonic calf raise exercises, has very mild tenderness on palpation of the Achilles tendon, and has been able to tolerate light running without a flare-up in tendon irritability and aggravation of symptoms.
For example, increasing duration under tension during heavy slow-resistance training may increase a strain on the tendon and result in greater adaptation, however increasing speed is going to be more likely to improve power and prepare for sporting activities involving the Stretch Shortening Cycle.
Exercises outlined is as follows:
- Double-leg hop
- Single leg hop
- Single leg step hops
- Hopscotch with activation band
Adjunct Therapies[edit | edit source]
In combination with approaches to optimize biomechanics and prescribe exercise therapy, adjunct therapies may be used. These forms of therapy usually cannot resolve or prevent injury, they are used more for symptom management.
Manual Therapy[edit | edit source]
There is no clinical evidence but there is expert level consensus to support the use of joint mobilizations in the acute stage if the assessment reveals joint restriction. There is a small amount of clinical evidence and more substantial expert level consensus to support the use of joint mobilizations in the chronic stage if the assessment reveals joint restriction[31] May consider using manual therapy after a comprehensive evaluation of the hip, knee, foot, and ankle reveals joint dysfunction. Ankle mobilizations can be used for dorsiflexion limitation of the talocrural joint and varus- or valgus limitation of the subtalar joint [41][5].
The effectiveness of deep cross frictions is not scientifically proven and gives limited results[24][42][43] There is a small amount of clinical evidence to support the use of soft tissue techniques, such as frictions, in the chronic stage. May consider a trial of soft tissue techniques, such as frictions, in the chronic stage[31].
Electrotherapy Modalities[edit | edit source]
There is conflicting evidence to support the use of Extracorporeal Shock Wave Therapy (ESWT) in the chronic stage. There is evidence suggesting that the outcomes are dependent upon the dosage of the shock wave energy (EFD ‐ energy flux density = mJ/mm²), rather than the type of shock wave generation (focused vs. radial ESWT). There is also evidence that the use of anaesthetic required in high energy protocols decreases the effectiveness of ESWT. Therefore, using low energy ESWT protocols without the need for anaesthetic are recommended as more practical, more tolerable, and less expensive with equivalent results. Low energy ESWT protocols can apply to both focused and radial ESWT. Consider a trial of ESWT in the chronic stage, especially if other interventions have failed, at the following parameters:
- Low energy SWT: EFD = 0.18 – 0.3 mJ/mm² (2‐4 Bars)
- 2000‐3000 shocks
- 15‐30 Hz
- 3‐5 sessions, weekly intervals.
There is no clinical evidence to support the use of Ultrasound and Low-Level Laser Therapy.
Iontophoresis[edit | edit source]
There is a small amount of evidence to support the application of iontophoresis using dexamethasone in the acute stage but not in the chronic stage. The role of iontophoresis is still investigational. May consider, in the acute stage, a trial of iontophoresis, 0.4% dexamethasone (aqueous), 80 mA‐min; 6 sessions over 3 weeks. A program of concentric‐eccentric exercises should be continued in combination with iontophoresis if exercise loading is tolerated.
Taping[edit | edit source]
Antipronation taping is supported by expert opinion, not clinical evidence. May consider using taping, possibly prior to Introduction to Orthotics in the acute stage[31]. Clinicians should not use therapeutic elastic tape to reduce pain or improve functional performance in patients with Achilles tendinopathy. Clinicians may use rigid taping to decrease strain on the Achilles tendon and/or alter foot posture in patients with Achilles tendinopathy.[23]
Night Splints[edit | edit source]
There is expert opinion to
- Support the use of night splints and braces in the acute stage
- Moderate amount of evidence against the use of night splints and braces in the chronic stage.
Consider a trial of night splints and braces in the acute stage but NOT using night splints and braces in the chronic stage in conjunction with exercise[31].
Dry Needling[edit | edit source]
Clinicians may use combined therapy of dry needling with an injection under ultrasound guidance and eccentric exercise to decrease pain for individuals with symptoms greater than 3 months and increased tendon thickness.[23]
Medical Management[edit | edit source]
Medication[edit | edit source]
Inflammation is necessary to start a restoration process in the damaged tissue, but the use of certain medications, such as corticosteroids and quinolones counter the inflammation, and as a result also the restoration process. Even when the patient does not take this medication, tendinopathy is also a consequence of a disrupted restoration process[44]
Corticosteroid Injections[edit | edit source]
Corticosteroid injection (CSI) appears to have short‐term pain‐relieving effects but no effect or detrimental effects in the longer term. The short‐term effect of CSI has been shown in the Achilles tendon with improvement in walking and reduction in tendon diameter as measured by ultrasonography. Intratendinous injection is contraindicated because of the catabolic effects, although a recent study of CSI into intratendinous vessels in six tendons has shown promising results. Peritendinous injection has fewer effects on the tendon and could be a worthwhile adjunct to a considered management program. CSI may be most beneficial when used to relieve pain while continuing to undertake an exercise program[45].
Sclerosering Injections[edit | edit source]
The role of neovascularization in tendon pain has been further examined in a pilot study where a vascular sclerosant (Polidocanol-an aliphatic non‐ionized nitrogen‐free substance with a sclerosing and anaesthetic effect) was injected in the area with neovascularization anterior to the tendon. A Short‐term (6 months) evaluation of this treatment showed that the clear majority were pain-free after a mean of two treatments. The tendons that were pain-free had no neovascularization either on the outside or the inside. A 2‐year follow‐up of these patients showed that the same eight patients remained pain-free with no vessels in the tendon. Ultrasonographically, tendon thickness had decreased and the structure looked rather normal [45].
Rehabilitation after a sclerosing injection consists of 1 - 3 days of rest; then tendon-loading activity increases gradually avoiding maximum loading. After 2 weeks tendon loading activity is allowed (jumping, fast runs, heaving strength training). This research suggests a clinical role for sclerosing therapy for those who fail to respond to eccentric exercise[45].
Platelet-Rich Plasma Injections[edit | edit source]
Research shows that injections of Platelet-Rich Plasma (PRP) during a period of 3 months in people with chronic Achilles Tendinopathy have no positive effect compared to placebo (saline). The only significant effect of PRP injections compared to placebo was a change in tendon thickness: this difference indicates that a PRP injection could increase tendon thickness compared with saline injection[45]
Minimal Invasive Procedures[edit | edit source]
According to a recent study, Minimal invasive procedure treats insertional Achilles tendinopathy, a common and chronic musculoskeletal disorder in which patients experience pain at the Achilles tendon.[46] The chronic degenerative condition is often painful for athletes who perform push-off activities, like basketball and soccer players. The key-hole procedure, referred to as percutaneous Zadek osteotomy (ZO), may significantly reduce pain and provide pain relief within six weeks after this procedure compared to 23 weeks for recovery post-traditional open surgery.
The short recovery period involves protecting the foot in a splint or walker boot for 2 weeks, then resuming weight-bearing while wearing a removable walker boot for an additional 4 weeks. Physiotherapy also begins two weeks after surgery. Athletes are allowed to return to shoes 6 weeks after the outpatient procedure.[46]
Operative Surgery[edit | edit source]
The aim of surgical treatment for tendinopathy is to irritate the tendon to initiate a chemically mediated reaction response. Surgery may consist of simple procedures, percutaneous tenotomy, open procedures, and removal of the infected tendon part. In 75% of subjects who underwent tenotomy experienced a positive result after 18 months. An open procedure of the Achilles tendon resulted in better outcomes of tendons without a focal lesion[45]
References[edit | edit source]
- ↑ Clain MR, Baxter DE. Achilles tendinitis. Foot & ankle. 1992 Oct;13(8):482-7.
- ↑ 2.0 2.1 Radioopedia Achilles Tendinopathy Available:https://radiopaedia.org/articles/achilles-tendinopathy (accessed 10.6.2022)
- ↑ Schubert TE, Weidler C, Lerch K, Hofstädter F, Straub RH. Achilles tendinosis is associated with sprouting of substance P positive nerve fibres. Annals of the rheumatic diseases. 2005 Jul 1;64(7):1083-6.
- ↑ Very well health The Health Benefits of Glycosaminoglycans Available: https://www.verywellhealth.com/glycosaminoglycans-5092414 (accessed 11.6.2022)
- ↑ 5.0 5.1 5.2 5.3 5.4 Wilson JJ, Best TM. Common overuse tendon problems: a review and recommendations for treatment. American family physician. 2005 Sep 1;72(5):811-8.
- ↑ 6.0 6.1 Paavola M, Kannus P, Järvinen TA, Khan K, Józsa L, Järvinen M. Achilles tendinopathy. JBJS. 2002 Nov 1;84(11):2062-76.
- ↑ Cook JL, Purdam CR. Is tendon pathology a continuum? A pathology model to explain the clinical presentation of load-induced tendinopathy. British journal of sports medicine. 2009 Jun 1;43(6):409-16.
- ↑ VAN DER, Arnt, and Liselot VAN DE WALLE. "Het excentrisch trainen van de kuitspieren."
- ↑ Radiopedia Paratenon Available:https://radiopaedia.org/articles/paratenon (accessed 10.6.2022)
- ↑ Kiewiet NJ, Holthusen SM, Bohay DR, Anderson JG. Gastrocnemius recession for chronic noninsertional Achilles tendinopathy. Foot & Ankle International. 2013 Apr;34(4):481-5.
- ↑ Ahmed IM, Lagopoulos M, McConnell P, Soames RW, Sefton GK. Blood supply of the Achilles tendon. Journal of orthopaedic research. 1998 Sep;16(5):591-6.
- ↑ Carr AJ, Norris SH. The blood supply of the calcaneal tendon. The Journal of bone and joint surgery. British volume. 1989 Jan;71(1):100-1.
- ↑ Young JS, Maffulli N. Etiology and epidemiology of achilles tendon problems. The Achilles Tendon. 2007 Sep 15:39-49.
- ↑ Gaida JE, Alfredson H, Kiss ZS, Bass SL, Cook JL. Asymptomatic Achilles tendon pathology is associated with a central fat distribution in men and a peripheral fat distribution in women: a cross sectional study of 298 individuals. BMC musculoskeletal disorders. 2010 Dec;11(1):1-9.
- ↑ Gaida JE, Ashe MC, Bass SL, Cook JL. Is adiposity an under‐recognized risk factor for tendinopathy? A systematic review. Arthritis Care & Research: Official Journal of the American College of Rheumatology. 2009 Jun 15;61(6):840-9.
- ↑ Mokone GG, Schwellnus MP, Noakes TD, Collins M. The COL5A1 gene and Achilles tendon pathology. Scandinavian journal of medicine & science in sports. 2006 Feb;16(1):19-26.
- ↑ September AV, Cook J, Handley CJ, van der Merwe L, Schwellnus MP, Collins M. Variants within the COL5A1 gene are associated with Achilles tendinopathy in two populations. British journal of sports medicine. 2009 May 1;43(5):357-65.
- ↑ Abate M, Schiavone C, Salini V, Andia I. Occurrence of tendon pathologies in metabolic disorders. Rheumatology. 2013 Apr 1;52(4):599-608.
- ↑ Ames PR, Longo UG, Denaro V, Maffulli N. Achilles tendon problems: not just an orthopaedic issue. Disability and rehabilitation. 2008 Jan 1;30(20-22):1646-50.
- ↑ Eckenrode BJ, Kietrys DM, Stackhouse SK. Pain sensitivity in chronic Achilles Tendinopathy. International Journal of Sports Physical Therapy. 2019 Dec;14(6):945.
- ↑ 21.0 21.1 21.2 21.3 Cook JL, Khan KM, Purdam C. Achilles tendinopathy. Manual therapy. 2002 Aug 1;7(3):121-30.
- ↑ 22.0 22.1 KL. Luscombe, P. S. (2003). Achilles tendinopathy. Trauma, 215-225.fckLR
- ↑ 23.0 23.1 23.2 23.3 23.4 23.5 23.6 Martin RL, Chimenti R, Cuddeford T, Houck J, Matheson JW, McDonough CM, Paulseth S, Wukich DK, Carcia CR. Achilles pain, stiffness, and muscle power deficits: midportion Achilles tendinopathy revision 2018: clinical practice guidelines linked to the International Classification of Functioning, Disability and Health From the Orthopaedic Section of the American Physical Therapy Association. Journal of Orthopaedic & Sports Physical Therapy. 2018 May;48(5):A1-38.
- ↑ 24.0 24.1 Hammer WI, editor. Functional soft-tissue examination and treatment by manual methods. Jones & Bartlett Learning; 2007.
- ↑ Wilson JJ, Best TM. Common overuse tendon problems: a review and recommendations for treatment. American family physician. 2005 Sep 1;72(5):811-8.
- ↑ Shibuya N, Thorud JC, Agarwal MR, Jupiter DC. Is calcaneal inclination higher in patients with insertional Achilles tendinosis? A case-controlled, cross-sectional study. The Journal of foot and ankle surgery. 2012 Nov 1;51(6):757-61.
- ↑ Royal London Hospital Test (CR)CRTechnologies. Available form https://www.youtube.com/watch?v=nM3yu_TR4H8&t=2s
- ↑ CRTechnologies.Arc SIgn Achilles (CR). Available form https://www.youtube.com/watch?v=zhP0CAsQr7U&t=24s
- ↑ Fouré A. New imaging methods for non-invasive assessment of mechanical, structural, and biochemical properties of human Achilles tendon: a mini review. Frontiers in physiology. 2016 Jul 27;7:324.
- ↑ Robinson JM, Cook JL, Purdam C, Visentini PJ, Ross J, Maffulli N, Taunton JE, Khan KM. The VISA-A questionnaire: a valid and reliable index of the clinical severity of Achilles tendinopathy. British journal of sports medicine. 2001 Oct 1;35(5):335-41.
- ↑ 31.0 31.1 31.2 31.3 31.4 31.5 BC Physical Therapy Tendinopathy Task Force: Dr. Joseph Anthony, Allison Ezzat, Diana Hughes, JR Justesen, Dr. Alex Scott, Michael Yates, Alison Hoens. Achilles Tendinopathy Toolkit. A Physical Therapy Knowledge Broker project supported by: UBC Department of Physical Therapy, Physiotherapy Associaton of BC, Vancouver Coastal Research Institute and Providence Healthcare Research Institute. 2012
- ↑ Scott A, Huisman E, Khan K. Conservative treatment of chronic Achilles tendinopathy. CMAJ. 2011 Jul 12;183(10):1159-65.
- ↑ Rio E, Kidgell D, Moseley GL, Gaida J, Docking S, Purdam C, Cook J. Tendon neuroplastic training: changing the way we think about tendon rehabilitation: a narrative review. British journal of sports medicine. 2016 Feb 1;50(4):209-15.
- ↑ 34.0 34.1 Malliaras P, Barton CJ, Reeves ND, Langberg H. Achilles and patellar tendinopathy loading programmes. Sports medicine. 2013 Apr 1;43(4):267-86.
- ↑ Beyer R, Kongsgaard M, Hougs Kjær B, Øhlenschlæger T, Kjær M, Magnusson SP. Heavy slow resistance versus eccentric training as treatment for Achilles tendinopathy: a randomized controlled trial. The American journal of sports medicine. 2015 Jul;43(7):1704-11.
- ↑ Cook JL, Purdam CR. The challenge of managing tendinopathy in competing athletes. British journal of sports medicine. 2014 Apr 1;48(7):506-9.
- ↑ Single leg heel raise isometric hold available from https://www.youtube.com/watch?v=7Udb4E3Uks8
- ↑ Soleus calf raises-seated available from https://www.youtube.com/watch?v=C_UwdAuD7ZY
- ↑ Single leg heel raise isometric hold available from https://www.youtube.com/watch?v=qW2XAz8hYf0&feature=emb_logo
- ↑ Achilles Tendonitis Rehab Phase 3 available from https://www.youtube.com/watch?v=t9IwvBjQA8I
- ↑ Scott A, Huisman E, Khan K. Conservative treatment of chronic Achilles tendinopathy. CMAJ. 2011 Jul 12;183(10):1159-65.
- ↑ Stasinopoulos D, Stasinopoulos I. Comparison of effects of exercise programme, pulsed ultrasound and transverse friction in the treatment of chronic patellar tendinopathy. Clinical rehabilitation. 2004 Jun;18(4):347-52.
- ↑ Joseph MF, Taft K, Moskwa M, Denegar CR. Deep friction massage to treat tendinopathy: a systematic review of a classic treatment in the face of a new paradigm of understanding. Journal of sport rehabilitation. 2012 Nov 1;21(4):343-53.
- ↑ Paavola M, Kannus P, Järvinen TA, Khan K, Józsa L, Järvinen M. Achilles tendinopathy. JBJS. 2002 Nov 1;84(11):2062-76.
- ↑ 45.0 45.1 45.2 45.3 45.4 Alfredson, Håkan, and Jill Cook. "A treatment algorithm for managing Achilles tendinopathy: new treatment options." British journal of sports medicine 41.4 (2007): 211-216.
- ↑ 46.0 46.1 Silbernagel KG, Hanlon S, Sprague A. Current Clinical Concepts: Conservative Management of Achilles Tendinopathy. Journal of Athletic Training. 2020 May;55(5):438-47.